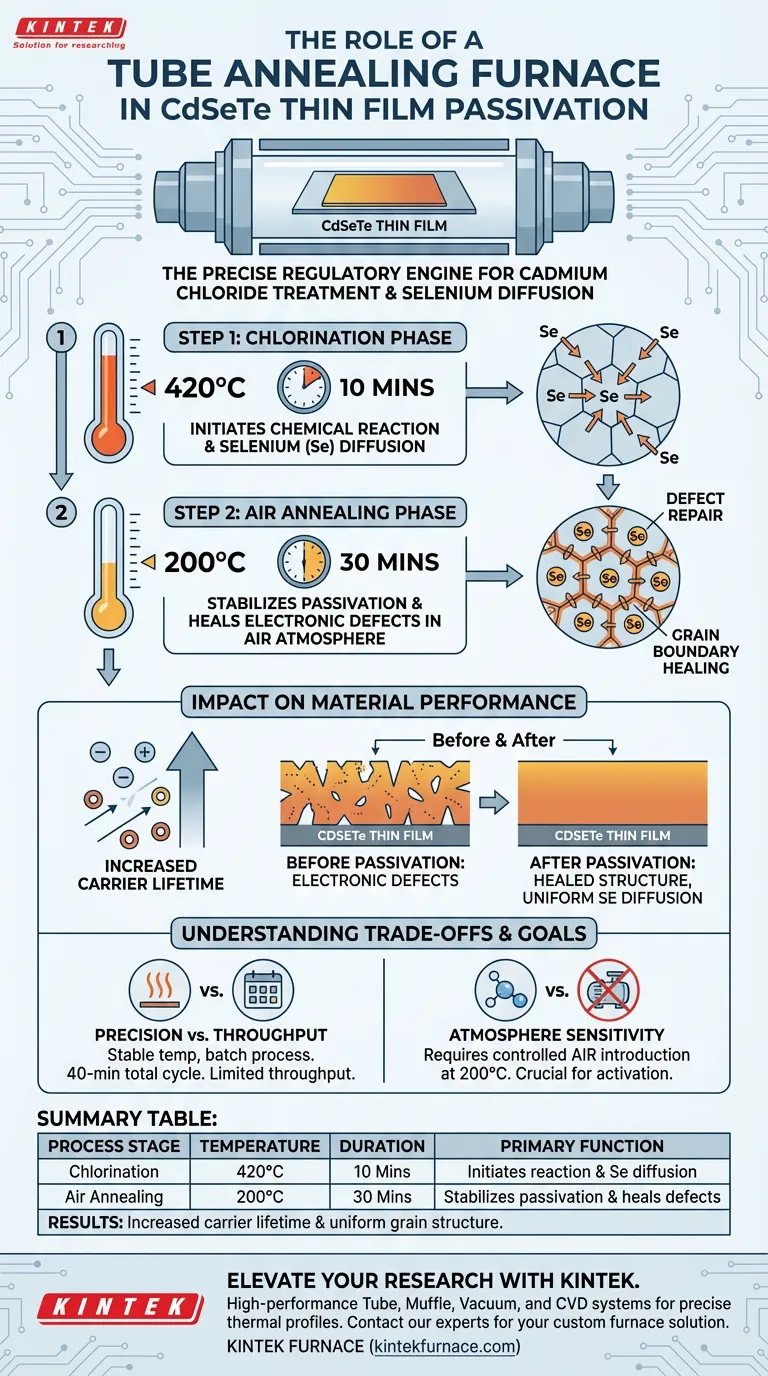
The tube annealing furnace serves as the precise regulatory engine for the Cadmium Chloride (CdCl2) treatment of CdSeTe thin films. It creates a strictly controlled thermal environment that executes a critical two-step heating process, driving the chemical changes required to passivate the material.
The furnace's primary function is to enable the diffusion of Selenium (Se) atoms along grain boundaries and into grain interiors. By maintaining a stable temperature field, it activates passivation mechanisms that significantly increase the carrier lifetime of the thin film.

The Mechanics of the Passivation Process
The Two-Step Thermal Profile
The effectiveness of the passivation relies on a specific thermal recipe that the tube furnace must execute with high precision.
First, the sample undergoes chlorination at 420°C for 10 minutes. This high-temperature phase initiates the chemical reaction between the film and the chloride treatment.
Subsequently, the furnace shifts to air annealing at 200°C for 30 minutes. This secondary stage is crucial for stabilizing the changes and finalizing the treatment.
Driving Selenium Diffusion
The core value of the tube furnace lies in its ability to drive atomic movement. The thermal energy provided forces Selenium (Se) atoms to migrate.
This diffusion occurs along the grain boundaries and penetrates into the grain interiors. This migration is the physical mechanism that repairs defects within the crystal structure.
The Impact on Material Performance
Activating Passivation
Passivation is not automatic; it requires an "activation energy" provided by the furnace.
By facilitating the specific diffusion of Selenium, the furnace effectively "heals" the electronic defects that would otherwise trap charge carriers.
Increasing Carrier Lifetime
The ultimate metric for success in this process is carrier lifetime—how long an electron or hole exists before recombining.
The stable temperature field ensures that the Se diffusion is uniform. This uniformity leads to a consistent increase in carrier lifetime across the film, which is essential for high-performance semiconductor devices.
Understanding the Trade-offs
Precision vs. Throughput
Tube furnaces excel at providing a stable temperature field and a sealed environment, which is vital for the delicate diffusion of Se atoms.
However, this precision often requires batch processing rather than continuous flow. The strict requirement for a 40-minute total process time (excluding ramp rates) means throughput is limited by the physics of the reaction.
Atmosphere Sensitivity
While some annealing processes require vacuum or inert gases (like Argon) to prevent oxidation, this specific CdSeTe process requires an air annealing phase.
Operators must ensure the furnace allows for the controlled introduction of air during the second stage (200°C). Failing to switch atmospheres correctly can prevent the final activation of the film.
Making the Right Choice for Your Goal
To maximize the potential of your CdSeTe thin films, align your furnace operations with your specific objectives:
- If your primary focus is Defect Reduction: Prioritize the stability of the 420°C stage to ensure maximum Selenium diffusion into the grain interiors.
- If your primary focus is Carrier Lifetime: Ensure the 200°C air annealing phase is strictly timed, as this stabilizes the passivation activated in the first step.
The tube furnace is not just a heater; it is the instrument that orchestrates the atomic diffusion necessary for high-quality CdSeTe devices.
Summary Table:
| Process Stage | Temperature | Duration | Primary Function |
|---|---|---|---|
| Chlorination | 420°C | 10 Minutes | Initiates chemical reaction and Se diffusion |
| Air Annealing | 200°C | 30 Minutes | Stabilizes passivation and heals electronic defects |
| Results | N/A | Total: 40 Mins | Increased carrier lifetime and uniform grain structure |
Elevate Your Semiconductor Research with KINTEK
Precision is non-negotiable when optimizing carrier lifetime in CdSeTe thin films. Backed by expert R&D and manufacturing, KINTEK offers high-performance Tube, Muffle, Vacuum, and CVD systems designed to deliver the stable temperature fields required for critical passivation treatments.
Whether you need customizable thermal profiles for Selenium diffusion or specialized atmospheric controls for air annealing, our laboratory high-temp furnaces are built to meet your unique semiconductor fabrication needs.
Ready to achieve superior material performance? Contact our experts today to find the perfect furnace solution for your lab.
Visual Guide
References
- Bérengère Frouin, Stéphane Collin. Quantitative assessment of selenium diffusion and passivation in CdSeTe solar cells probed by spatially resolved cathodoluminescence. DOI: 10.1063/5.0195398
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Custom Made Versatile CVD Tube Furnace Chemical Vapor Deposition CVD Equipment Machine
- Split Chamber CVD Tube Furnace with Vacuum Station CVD Machine
- Multi Heating Zones CVD Tube Furnace Machine for Chemical Vapor Deposition Equipment
- Magnesium Extraction and Purification Condensing Tube Furnace
- Slide PECVD Tube Furnace with Liquid Gasifier PECVD Machine
People Also Ask
- What is a CVD tube furnace and what is its primary function? Discover Precision Thin-Film Deposition
- Why are CVD tube furnace sintering systems indispensable for 2D material research and production? Unlock Atomic-Scale Precision
- What temperature range do standard CVD tube furnaces operate at? Unlock Precision for Your Material Deposition
- What improvements can be made to the bonding force of gate dielectric films using a CVD tube furnace? Enhance Adhesion for Reliable Devices
- What is the working principle of a CVD tube furnace? Achieve Precise Thin Film Deposition for Your Lab

















