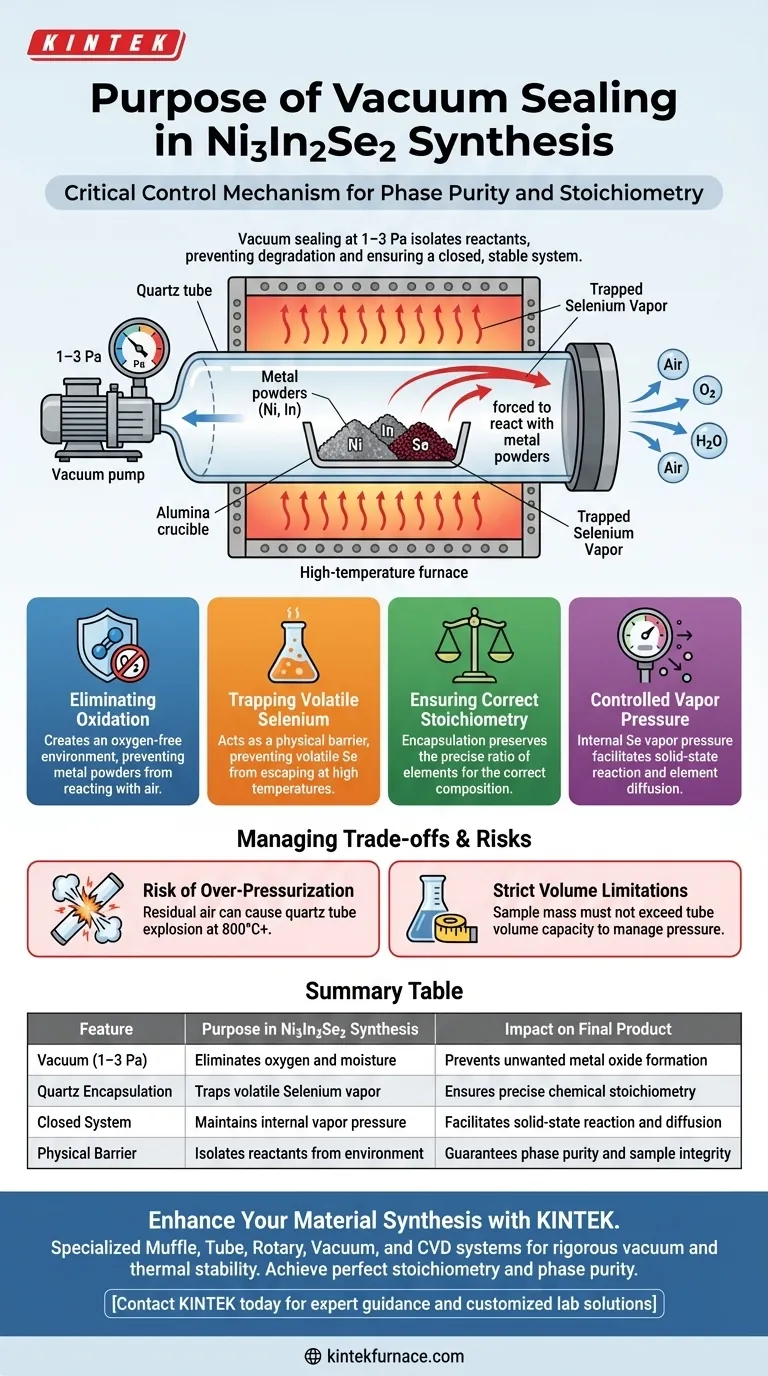
Vacuum sealing is the critical control mechanism that enables the successful synthesis of Ni3In2Se2 by isolating reactants from the external environment. Specifically, sealing the alumina crucible containing raw materials within a quartz tube at low pressures (1–3 Pa) prevents the degradation of raw materials and ensures the chemical reaction occurs within a closed, stable system.
By establishing an oxygen-free environment, this process preserves the precise chemical balance (stoichiometry) of the compound. It simultaneously prevents the oxidation of metal powders and traps volatile Selenium vapor, forcing it to react rather than escape.

Controlling the Chemical Atmosphere
To achieve phase purity in solid-state synthesis, you must strictly control the atmosphere surrounding the reactants.
Eliminating Oxidation
The primary function of the vacuum seal is to create an oxygen-free environment.
Metals used in this synthesis are highly susceptible to oxidation at high temperatures. If exposed to air during the heating process, the metal powders would react with oxygen rather than with the Selenium.
Protecting the Reactants
Vacuum sealing at 1–3 Pa removes air and moisture that could introduce impurities.
This ensures that the Nickel (Ni) and Indium (In) remain in their metallic state until they react with the Selenium, preventing the formation of unwanted metal oxides.
Managing Volatility and Stoichiometry
The most challenging aspect of synthesizing compounds like Ni3In2Se2 is managing elements that turn into gas (vaporize) at low temperatures.
Trapping Volatile Selenium
Selenium is a volatile element that vaporizes easily at the high temperatures required for synthesis.
Without a sealed environment, Selenium vapor would escape the crucible and exit the system entirely. The quartz tube acts as a physical barrier, trapping the Selenium vapor inside the reaction zone.
Ensuring Correct Stoichiometry
The loss of Selenium would ruin the stoichiometry (the precise ratio of elements) of the final product.
By encapsulating the reactants, you ensure that the specific amount of Selenium weighed at the start remains available for the reaction, leading to the correct Ni3In2Se2 composition.
Controlled Vapor Pressure
The sealed tube does more than just contain the material; it creates a pressurized environment.
As the Selenium vaporizes, it creates an internal vapor pressure that facilitates the solid-state reaction, helping the elements diffuse and combine correctly.
Understanding the Trade-offs
While vacuum sealing is essential for purity, it introduces specific physical risks that must be managed.
The Risk of Over-Pressurization
A sealed system operates under high internal pressure once heated.
If the initial vacuum is insufficient and residual air remains, the expansion of that gas at 800°C+ can cause the quartz tube to explode.
Strict Volume Limitations
Because of the pressure generated by the volatile Selenium, the size of the quartz tube relative to the sample mass is critical.
You cannot overfill the tube; there must be sufficient volume to accommodate the vapor pressure without breaching the quartz integrity.
Making the Right Choice for Your Goal
Vacuum sealing is not merely a storage technique; it is an active parameter in the synthesis process.
- If your primary focus is Phase Purity: Ensure the vacuum level is strictly maintained at or below 1–3 Pa to completely eliminate oxygen and moisture.
- If your primary focus is Stoichiometry: Prioritize the integrity of the seal and the thickness of the quartz wall to prevent any loss of Selenium vapor during the high-temperature dwell.
Success in synthesizing Ni3In2Se2 relies on treating the vacuum seal as a chemical reagent—it is just as vital to the reaction as the metal powders themselves.
Summary Table:
| Feature | Purpose in Ni3In2Se2 Synthesis | Impact on Final Product |
|---|---|---|
| Vacuum (1–3 Pa) | Eliminates oxygen and moisture | Prevents unwanted metal oxide formation |
| Quartz Encapsulation | Traps volatile Selenium vapor | Ensures precise chemical stoichiometry |
| Closed System | Maintains internal vapor pressure | Facilitates solid-state reaction and diffusion |
| Physical Barrier | Isolates reactants from environment | Guarantees phase purity and sample integrity |
Enhance Your Material Synthesis with KINTEK
Precision in Ni3In2Se2 synthesis starts with the right equipment. Backed by expert R&D and manufacturing, KINTEK offers specialized Muffle, Tube, Rotary, Vacuum, and CVD systems designed to maintain the rigorous vacuum levels and thermal stability required for your laboratory. Whether you need a standard setup or a customizable high-temperature furnace for unique research needs, our solutions empower you to achieve perfect stoichiometry and phase purity.
Ready to optimize your solid-state synthesis? Contact KINTEK today for expert guidance and customized lab solutions!
Visual Guide
References
- Yi Zhou. The Preparation and Physical Properties Study of the Kagome Lattice Semimetal Ni3In2Se2. DOI: 10.47297/taposatwsp2633-456926.20250604
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Laboratory Quartz Tube Furnace RTP Heating Tubular Furnace
- Multi Zone Laboratory Quartz Tube Furnace Tubular Furnace
- Vertical Laboratory Quartz Tube Furnace Tubular Furnace
- 1200℃ Split Tube Furnace Laboratory Quartz Tube Furnace with Quartz Tube
- High Pressure Laboratory Vacuum Tube Furnace Quartz Tubular Furnace
People Also Ask
- What role does a laboratory tube furnace system play in the catalytic pyrolysis of LLDPE? Enhancing Yield and Precision
- What conditions do tube furnaces provide for Au-Seeded TiO2 nanowires? Master Precision Thermal Synthesis
- What role does a laboratory tube furnace play in the carbonization process of porous carbon particles? Expert Insights
- How does a laboratory tube furnace regulate the positioning of platinum (Pt) atoms? Mastering Atomic-Scale Engineering
- What conditions does a tube furnace provide for post-ion-implantation? Achieve Precise Microstructural Repair
















