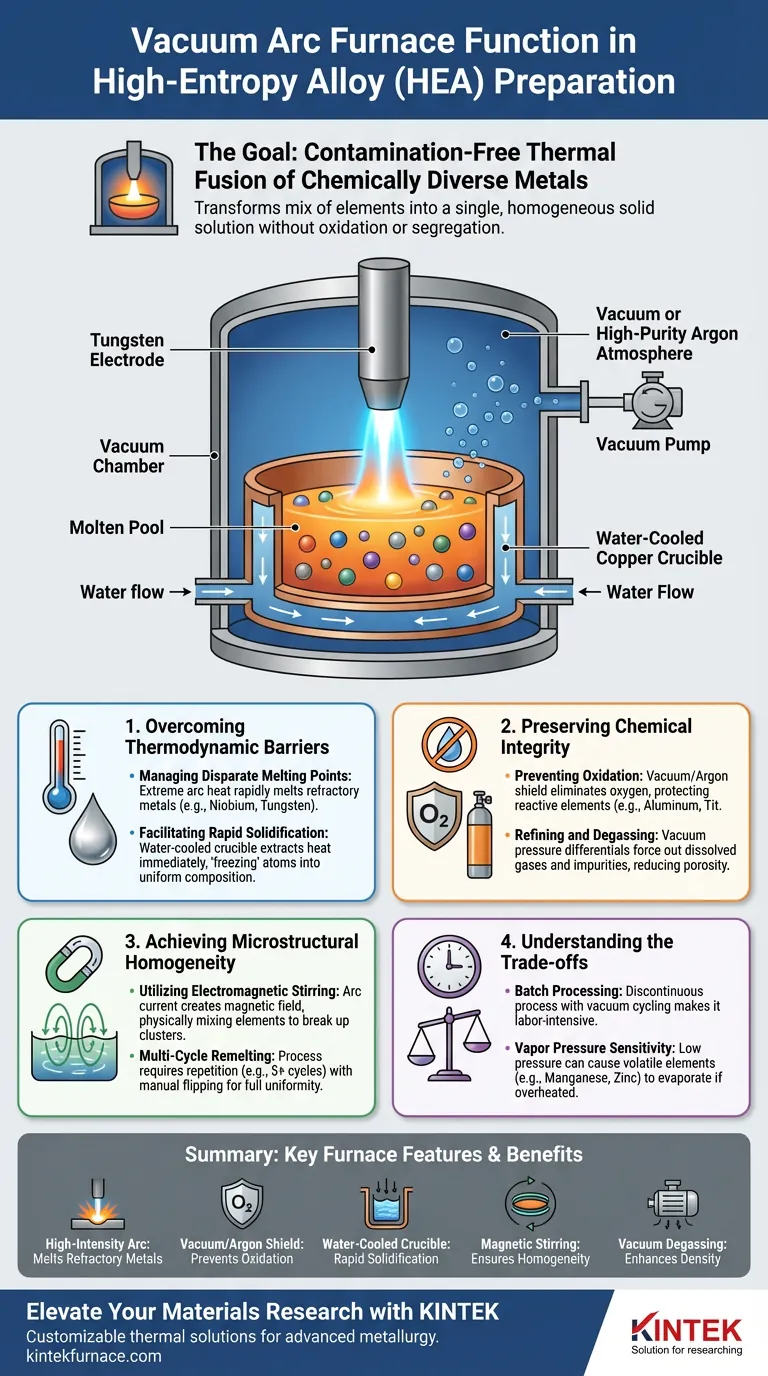
The primary function of a vacuum arc furnace in preparing high-entropy alloy (HEA) ingots is to provide an extreme, contamination-free thermal environment that forces chemically diverse metals to fuse completely. By generating high-temperature electric arcs within a vacuum or high-purity argon atmosphere, the furnace rapidly melts elements with vastly different melting points while preventing the oxidation of reactive components.
Core Takeaway While the furnace’s mechanism is thermal, its strategic value is chemical stability and structural uniformity. It is the critical processing step that transforms a mix of refractory and reactive elements into a single, homogeneous solid solution without the defects caused by oxidation or elemental segregation.

Overcoming Thermodynamic Barriers
The central challenge in creating high-entropy alloys is combining elements that naturally resist mixing due to drastic differences in physical properties.
Managing Disparate Melting Points
High-entropy alloys often combine refractory metals (like Niobium or Tungsten) with lower-melting-point metals.
The vacuum arc furnace utilizes non-consumable electrodes (typically Tungsten) to generate a localized, high-intensity electric arc. This provides the extreme heat necessary to melt the refractory components rapidly.
This rapid melting ensures that high-melting-point elements fuse effectively before the lower-melting-point elements can evaporate or separate significantly.
Facilitating Rapid Solidification
The furnace operates using a water-cooled copper crucible.
Once the arc is extinguished, the contact with the water-cooled surface extracts heat immediately from the molten pool.
This leads to rapid solidification, which helps "freeze" the atoms in place, creating an initial ingot with a uniform composition and preventing the components from separating back into their individual phases.
Preserving Chemical Integrity
Beyond temperature, the atmospheric control within the furnace is the defining factor for ingot quality.
Preventing Oxidation of Active Elements
Many HEAs utilize reactive elements such as Aluminum, Titanium, and Zirconium, which oxidize instantly in standard air.
The furnace creates a high-vacuum environment (often around $3 \times 10^{-3}$ Pa) or fills the chamber with high-purity inert gas (Argon).
This protective atmosphere eliminates oxygen, preventing the formation of brittle oxides and ensuring that active elements are retained in the metallic matrix rather than being lost as slag or dross.
Refining and Degassing
The vacuum environment utilizes pressure differentials to purify the melt.
Dissolved gases (like hydrogen) and impurities are forced out of the liquid metal and removed by the vacuum system.
This significantly reduces porosity and inclusions, resulting in a denser, higher-performance alloy structure.
Achieving Microstructural Homogeneity
Melting the metals once is rarely sufficient for HEAs; the furnace facilitates processes to ensure the alloy is uniform throughout.
Utilizing Electromagnetic Stirring
The electric current used to generate the arc creates a magnetic field.
This field induces a stirring motion within the molten pool, physically mixing the elements.
This electromagnetic stirring is crucial for breaking up clusters of heavy elements that might otherwise sink to the bottom of the crucible.
The Necessity of Multi-Cycle Remelting
To eliminate elemental segregation completely, the process requires repetition.
Operators manually flip the solidified ingot and remelt it multiple times—often at least five cycles.
This leverages the furnace's capabilities to ensure that even small-scale ingots (e.g., 100 grams) achieve chemical uniformity at the microscale.
Understanding the Trade-offs
While the vacuum arc furnace is the standard for high-purity ingots, it requires specific operational protocols to be effective.
Batch Processing Limitations
The process is inherently discontinuous. The requirement to break vacuum, flip the ingot, and re-establish the atmosphere for multiple remelting cycles makes it labor-intensive compared to continuous casting methods.
Sensitivity to Vapor Pressure
While the vacuum prevents oxidation, the low pressure can cause elements with high vapor pressures (like Manganese or Zinc) to evaporate if the melt is overheated. Careful control of the argon pressure is required to balance oxidation prevention with composition retention.
Making the Right Choice for Your Goal
To maximize the utility of a vacuum arc furnace for your specific alloy system, focus on these operational priorities:
- If your primary focus is Compositional Accuracy: Prioritize the use of a high-purity argon backfill rather than a deep vacuum to suppress the evaporation of volatile elements while still preventing oxidation.
- If your primary focus is Microstructural Uniformity: Commit to a minimum of five remelting cycles with manual flipping between each to fully leverage electromagnetic stirring and eliminate segregation.
Ultimately, the vacuum arc furnace acts as a high-precision equalizer, forcing incompatible elements into a unified, high-purity material state.
Summary Table:
| Feature | Function in HEA Preparation | Benefit |
|---|---|---|
| High-Intensity Arc | Rapidly melts refractory metals (e.g., Tungsten, Niobium) | Ensures complete fusion of disparate elements |
| Vacuum/Argon Shield | Eliminates oxygen and nitrogen interaction | Prevents oxidation of reactive elements like Al and Ti |
| Water-Cooled Crucible | Facilitates rapid solidification | Minimizes elemental segregation and 'freezes' structure |
| Magnetic Stirring | Induces physical mixing within the molten pool | Breaks up heavy element clusters for homogeneity |
| Vacuum Degassing | Removes dissolved gases and impurities | Reduces porosity and enhances alloy density |
Elevate Your Materials Research with KINTEK
Precision is the heartbeat of high-entropy alloy development. KINTEK provides industry-leading thermal solutions, including Vacuum, CVD, Muffle, and Tube furnaces, specifically engineered to meet the rigorous demands of advanced metallurgy.
Backed by expert R&D and world-class manufacturing, our systems are fully customizable to handle your unique refractory and reactive metal requirements. Whether you are aiming for microstructural homogeneity or contamination-free processing, KINTEK delivers the reliability you need to push the boundaries of material science.
Ready to refine your melting process? Contact us today to discuss your custom furnace needs with our engineering team!
Visual Guide
References
- Yukun Lv, Jian Chen. Improving Mechanical Properties of Co-Cr-Fe-Ni High Entropy Alloy via C and Mo Microalloying. DOI: 10.3390/ma17020529
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Vacuum Hot Press Furnace Machine Heated Vacuum Press Tube Furnace
- Vacuum Sealed Continuous Working Rotary Tube Furnace Rotating Tube Furnace
- Vacuum Hot Press Furnace Machine Heated Vacuum Press
- Vacuum Heat Treat Furnace with Ceramic Fiber Liner
- Laboratory Vacuum Tilt Rotary Tube Furnace Rotating Tube Furnace
People Also Ask
- How have vacuum hot press furnaces transformed material processing? Achieve Superior Density and Purity
- What are the benefits of the vacuum environment system in a vacuum hot press furnace? Unlock High-Density Sintering
- How does the uniaxial pressure applied by a vacuum hot press furnace affect the microstructure of ZrC-SiC materials?
- What is the significance of the vacuum environment for sintering stainless steel? Unlock High-Density Purity
- What is the primary function of the vacuum environment in a vacuum hot press furnace during titanium alloy processing? Prevent Embrittlement for Superior Ductility



















