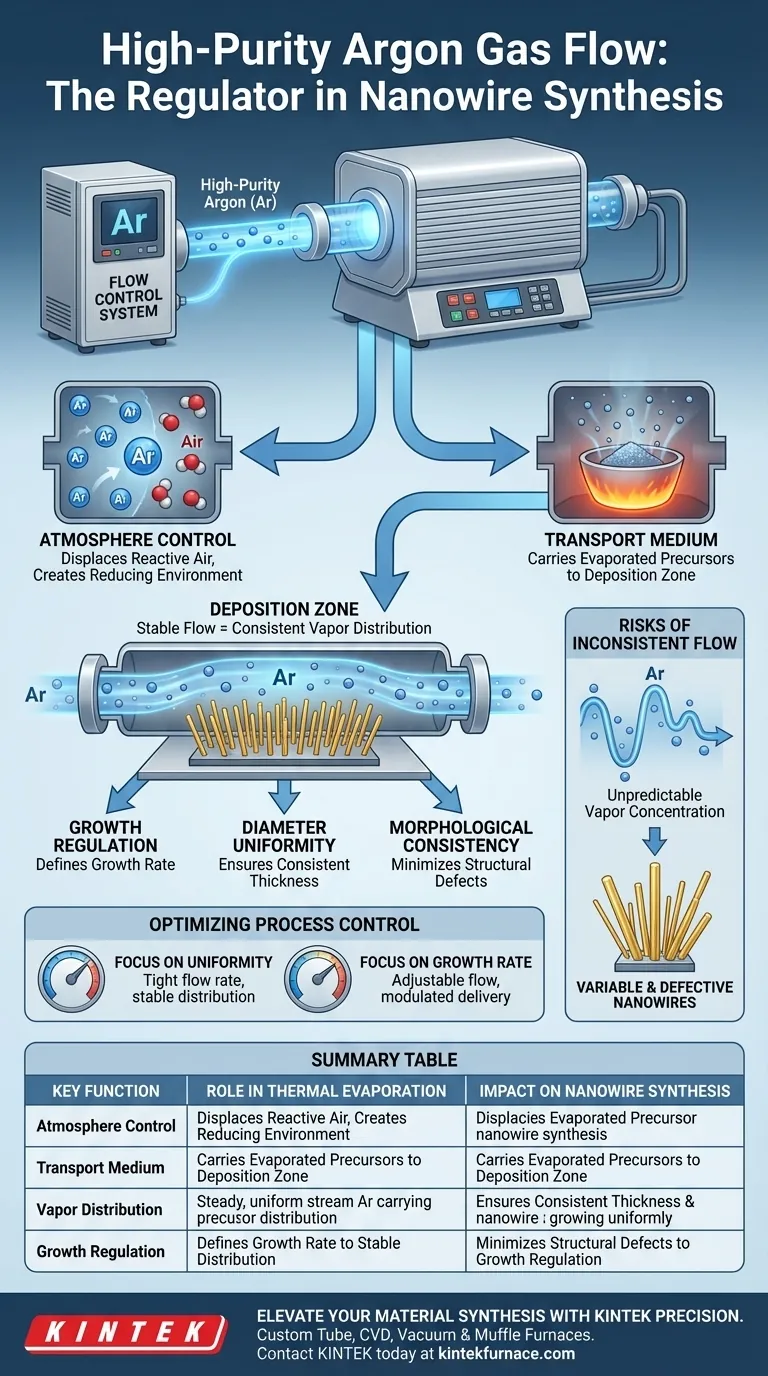
The high-purity Argon (Ar) gas flow control system serves as the critical regulator in thermal evaporation, functioning simultaneously as an environmental stabilizer and a transport vehicle. Its primary job is to displace reactive air to create a reducing environment while physically carrying evaporated precursor molecules to the deposition zone for nanowire synthesis.
Core Takeaway The stability of your Argon flow is the deciding factor in the structural quality of your nanowires. By dictating the concentration distribution of vapor components, the flow control system directly governs the growth rate, diameter uniformity, and morphological consistency of the final Si:B nanowires.

Establishing the Reaction Environment
Displacing Reactive Air
The first function of the Argon system is to manage the atmosphere within the furnace tube. By introducing high-purity gas, the system displaces ambient air to maintain a reducing environment. This prevents unwanted reactions, such as oxidation, that would compromise the purity of the synthesis.
Acting as a Transport Medium
Beyond environmental control, Argon acts as an inert carrier gas. Once the precursor materials evaporate, the Argon flow physically transports these gaseous molecules from the source to the deposition zone where growth occurs.
The Link Between Flow and Morphology
Regulating Concentration Distribution
The precision of the flow control system determines how vapor components are distributed throughout the tube. Stable flow control ensures that the concentration of these vapors remains consistent across the synthesis zone.
Defining Nanowire Characteristics
This concentration stability directly impacts the physical properties of the resulting Si:B nanowires. The flow rate dictates the growth rate, ensuring the wires develop at the intended speed.
Ensuring Structural Uniformity
Furthermore, the flow system controls the diameter distribution and morphological consistency. A stable flow results in wires that are uniform in thickness and shape, which is essential for predictable material performance.
The Risks of Inconsistent Flow
Impact on Product Quality
If the flow control system lacks stability, the vapor concentration in the deposition zone will fluctuate. This inconsistency leads to unpredictable variations in nanowire diameter and structural defects. Without precise regulation, achieving morphological consistency becomes impossible, resulting in a batch of nanowires with highly variable physical traits.
Optimizing Process Control
To achieve specific synthesis goals, you must treat the gas flow as a primary variable.
- If your primary focus is Uniformity: Prioritize absolute stability in the flow rate to maintain a constant vapor concentration, which ensures a tight diameter distribution.
- If your primary focus is Growth Rate: Adjust the flow to modulate the speed at which precursor molecules are delivered to the deposition zone.
Mastering the Argon flow is effectively mastering the geometry and consistency of your nanowires.
Summary Table:
| Key Function | Role in Thermal Evaporation | Impact on Nanowire Synthesis |
|---|---|---|
| Atmosphere Control | Displaces reactive air & creates a reducing environment | Prevents oxidation; ensures material purity |
| Transport Medium | Carries evaporated precursor molecules to deposition zone | Governs delivery speed and growth location |
| Vapor Distribution | Regulates concentration of gaseous components | Ensures diameter uniformity & morphological consistency |
| Growth Regulation | Maintains stable flow rates throughout the process | Defines growth rate and minimizes structural defects |
Elevate Your Material Synthesis with KINTEK Precision
Achieving consistent nanowire morphology requires absolute control over your thermal environment. Backed by expert R&D and manufacturing, KINTEK provides high-performance Tube, CVD, Vacuum, and Muffle furnaces designed to handle sophisticated gas flow requirements.
Whether you are synthesizing Si:B nanowires or developing advanced nanomaterials, our systems are fully customizable to meet your unique research specifications. Ensure your next batch meets the highest standards of uniformity and purity.
Contact KINTEK today to discuss your high-temperature furnace needs!
Visual Guide
References
- Feng Yang, Shihua Zhao. Preparation and photoelectric properties of Si:B nanowires with thermal evaporation method. DOI: 10.1371/journal.pone.0316576
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- RF PECVD System Radio Frequency Plasma Enhanced Chemical Vapor Deposition
- Custom Made Versatile CVD Tube Furnace Chemical Vapor Deposition CVD Equipment Machine
People Also Ask
- What are the key advantages of PECVD technology? Achieve Low-Temperature, High-Quality Thin Film Deposition
- How is silicon dioxide deposited from tetraethylorthosilicate (TEOS) in PECVD? Achieve Low-Temperature, High-Quality SiO2 Films
- How are deposition rates and film properties controlled in PECVD? Master Key Parameters for Optimal Thin Films
- What is Plasma-enhanced chemical vapor deposition (PECVD) and its applications? Unlock Low-Temperature Thin-Film Coating
- What are the advantages of PECVD in film deposition? Achieve Low-Temp, High-Quality Coatings

