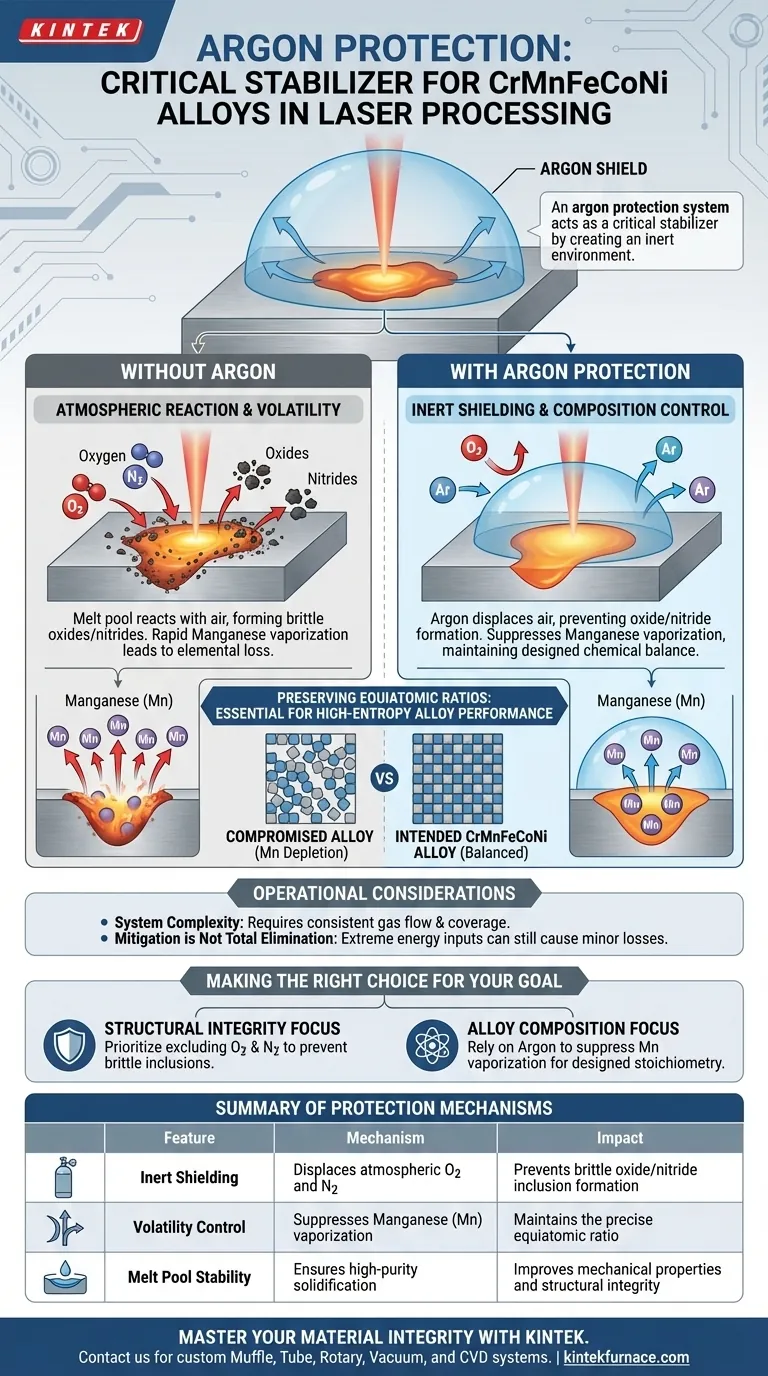
An argon protection system acts as a critical stabilizer for CrMnFeCoNi alloys during the intense heat of laser processing. By enveloping the processing zone in an inert gas, it prevents the molten metal from reacting with atmospheric oxygen or nitrogen. Simultaneously, it suppresses the vaporization of volatile elements like Manganese, ensuring the final material retains its intended chemical balance.
High-temperature laser processing threatens alloy integrity through immediate oxidation and selective elemental loss. An argon shield neutralizes these threats, preserving the precise equiatomic ratios required for high-performance high-entropy alloys.

The Mechanics of Atmospheric Shielding
Creating an Inert Environment
Laser processing generates ultra-high temperatures that render metals highly reactive.
Without protection, the melt pool would immediately interact with the surrounding air.
The argon system displaces this air, preventing the formation of oxides and nitrides that would otherwise compromise the alloy's mechanical properties.
Protecting the Melt Pool
The stability of the melt pool is paramount for consistent solidification.
By excluding Oxygen and Nitrogen, the argon shield ensures the liquid metal remains pure.
This allows the alloy to resolidify without inclusions or brittle phases caused by atmospheric contamination.
Managing Elemental Volatility
The Manganese Challenge
In the specific context of CrMnFeCoNi alloys, Manganese (Mn) presents a unique challenge due to its low boiling point.
Under the extreme heat of a laser, Mn is prone to rapid vaporization.
If left unchecked, this leads to a significant depletion of Manganese in the final structure, altering the alloy's fundamental characteristics.
Preserving Equiatomic Ratios
High-entropy alloys rely on specific atomic ratios—often equiatomic—to achieve their superior properties.
The argon protection system helps mitigate the preferential loss of volatile elements like Mn.
By keeping the vaporization in check, the system ensures the remelted region remains as close as possible to the designed chemical composition.
Operational Considerations and Trade-offs
System Complexity vs. Material Quality
While an argon system is essential for quality, it introduces complexity to the manufacturing setup.
Operators must ensure consistent gas flow and coverage; any turbulence or gaps in the shield can lead to localized defects.
Mitigation is Not Total Elimination
It is important to note that while argon helps to mitigate vaporization, it may not completely eliminate it under all processing parameters.
Extreme energy inputs could still lead to minor elemental losses, requiring careful calibration of laser power alongside the gas protection.
Making the Right Choice for Your Goal
To maximize the effectiveness of your laser processing setup, align your approach with your specific metallurgical targets:
- If your primary focus is Structural Integrity: Prioritize the argon shield's ability to exclude Oxygen and Nitrogen, which prevents the formation of brittle oxide inclusions.
- If your primary focus is Alloy Composition: rely on the argon environment to suppress the vaporization of Manganese, ensuring your final part matches the designed CrMnFeCoNi stoichiometry.
The argon protection system is not merely an accessory; it is a fundamental requirement for translating a high-entropy alloy design into a physical reality.
Summary Table:
| Feature | Protection Mechanism | Impact on CrMnFeCoNi Alloy |
|---|---|---|
| Inert Shielding | Displaces atmospheric O2 and N2 | Prevents brittle oxide/nitride inclusion formation |
| Volatility Control | Suppresses Manganese (Mn) vaporization | Maintains the precise equiatomic ratio |
| Melt Pool Stability | Ensures high-purity solidification | Improves mechanical properties and structural integrity |
| Thermal Regulation | Facilitates controlled cooling environment | Reduces localized defects and ensures consistency |
Master Your Material Integrity with KINTEK
Precision in high-entropy alloys requires more than just high temperatures; it demands total atmospheric control. KINTEK provides industry-leading Muffle, Tube, Rotary, Vacuum, and CVD systems designed to meet the rigorous demands of your R&D or manufacturing processes. Our customizable laboratory furnaces ensure that elements like Manganese remain stable and your alloys stay pure.
Ready to elevate your laser processing and thermal treatments? Contact KINTEK today for a custom solution
Visual Guide
References
- Ajay Talbot, Yu Zou. Laser Remelting of a CrMnFeCoNi High‐Entropy Alloy: Effect of Energy Density on Elemental Segregation. DOI: 10.1002/adem.202501194
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
People Also Ask
- What is Hot-filament Chemical Vapor Deposition (HFCVD)? Achieve High-Quality Thin Films with Precision Control
- What materials are commonly used in CVD coating systems? Explore Key Materials for Superior Coatings
- What is the significance of the vacuum pressure control system in the CVD coating process for powders?
- How can customers maximize the quality of CVD coatings? Master Pre-Coating Prep for Superior Results
- What is the function of high-purity Argon (Ar) gas during CVD? Optimize Your Thin Film Uniformity and Purity
