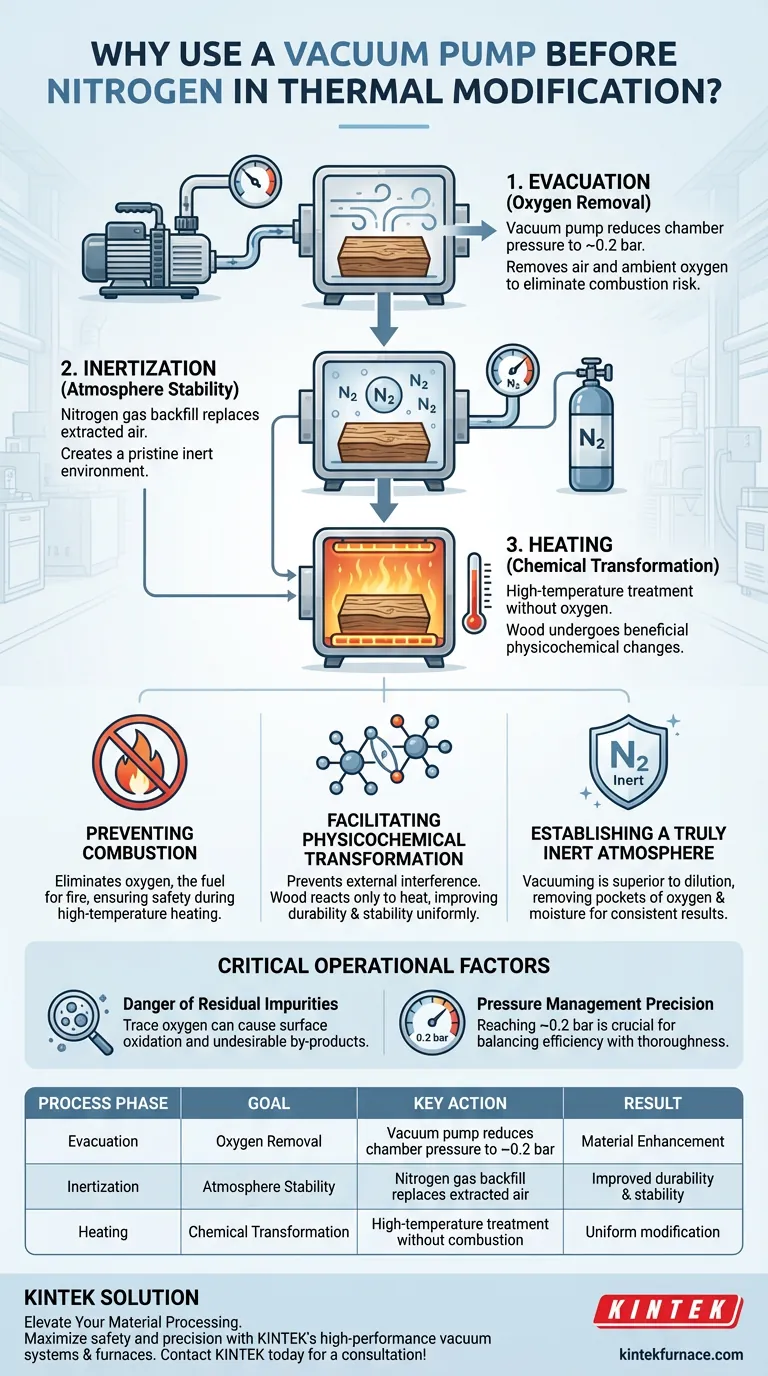
The primary purpose of using a vacuum pump in thermal modification is to completely eliminate oxygen from the treatment chamber before high temperatures are applied. By reducing the chamber pressure to a low level, typically around 0.2 bar, the system removes the ambient air that would otherwise react with the wood. This evacuation step is the prerequisite for establishing a purely inert atmosphere once nitrogen gas is introduced.
By evacuating air prior to nitrogen backfilling, operators eliminate the risk of combustion and unwanted oxidation. This creates the specific oxygen-free conditions required for the wood to undergo beneficial physicochemical changes rather than degradation.

Creating a Controlled Chemical Environment
Preventing Combustion at High Temperatures
The most immediate risk in thermal modification is fire. The process involves heating wood to temperatures where it would naturally burn if air were present.
Oxygen is the fuel for combustion. If the chamber contains residual air during the heating phase, the wood will ignite or char uncontrolledly.
The vacuum pump removes this risk by physically extracting the air, ensuring the flammability limit is never reached.
Facilitating Physicochemical Transformation
The goal of thermal modification is not just to dry the wood, but to alter its chemical structure to improve durability and stability.
This process is known as physicochemical transformation.
For this transformation to occur uniformly, the wood must be treated in an environment that prevents external chemical interference. A pre-evacuated, nitrogen-filled chamber ensures the wood reacts only to the heat, not to atmospheric gases.
Establishing a Truly Inert Atmosphere
Simply pumping nitrogen into a chamber full of air (dilution) is often insufficient for high-quality modification.
Dilution leaves pockets of oxygen and moisture that can lead to inconsistent results.
Using a vacuum pump to reach 0.2 bar ensures that the vast majority of atmospheric gases are removed before the inert gas is introduced. When nitrogen enters the vacuum, it fills the void completely, creating a pristine environment.
Critical Operational Factors
The Danger of Residual Impurities
Even trace amounts of oxygen can be detrimental. In sensitive processes, residual oxygen acts as a contaminant.
It causes surface oxidation, which creates undesirable by-products on the material.
While the primary reference focuses on wood, this principle applies broadly (as seen in CVD processes); removing air prevents impurities from compromising the material's microstructure or surface properties.
Pressure Management Precision
The effectiveness of this process relies on the depth of the vacuum.
Reaching a pressure of roughly 0.2 bar is a specific target designed to balance efficiency with thoroughness.
If the pump fails to reach this low pressure, the subsequent nitrogen backfill will be contaminated, negating the benefits of the inert gas.
Ensuring Process Integrity
If your primary focus is Safety:
- Prioritize vacuum depth to ensure oxygen levels are below the combustion threshold before heating begins.
If your primary focus is Material Quality:
- Ensure the transition from vacuum to nitrogen is seamless to prevent oxidation, which guarantees a consistent physicochemical transformation.
The vacuum phase is not merely a preparatory step; it is the fundamental control mechanism that separates successful modification from material destruction.
Summary Table:
| Process Phase | Goal | Key Action |
|---|---|---|
| Evacuation | Oxygen Removal | Vacuum pump reduces chamber pressure to ~0.2 bar |
| Inertization | Atmosphere Stability | Nitrogen gas backfill replaces extracted air |
| Heating | Chemical Transformation | High-temperature treatment without combustion risk |
| Result | Material Enhancement | Improved durability and stability through uniform modification |
Elevate Your Material Processing with KINTEK
Maximize the safety and precision of your thermal modification workflows. Backed by expert R&D and manufacturing, KINTEK offers high-performance vacuum systems, Muffle, Tube, Rotary, and CVD furnaces, all customizable to meet your unique laboratory or industrial needs. Whether you are aiming for perfect physicochemical transformations or specialized material synthesis, our technical experts are ready to provide the ideal solution.
Take the next step in process excellence—contact KINTEK today for a consultation!
Visual Guide
References
- Guntis Sosins, Jānis Zicāns. Water-Related Properties of Wood after Thermal Modification in Closed Process under Pressure in Nitrogen. DOI: 10.3390/f15010140
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Vacuum Hot Press Furnace Machine Heated Vacuum Press Tube Furnace
- 2200 ℃ Tungsten Vacuum Heat Treat and Sintering Furnace
- Small Vacuum Heat Treat and Tungsten Wire Sintering Furnace
- High Pressure Laboratory Vacuum Tube Furnace Quartz Tubular Furnace
- Vacuum Heat Treat Sintering Furnace with Pressure for Vacuum Sintering
People Also Ask
- Why is electromagnetic stirring necessary for NbC-Cr7C3@graphene in W18Cr4V steel? Boost Alloy Performance
- How does the choice of ceramic molds affect the results when preparing steel sample ingots? Ensure Maximum Sample Purity
- What is the purpose of bottom-entry argon injection? Enhance Lithium-ion Battery Safety & Purge Efficiency
- Why are synthesized CdS nanorods dried in a laboratory vacuum oven? Preserving Nanostructure and Chemical Integrity
- What is the function of vacuum & heating in aluminum degassing? Enhancing Composite Integrity & Density



















