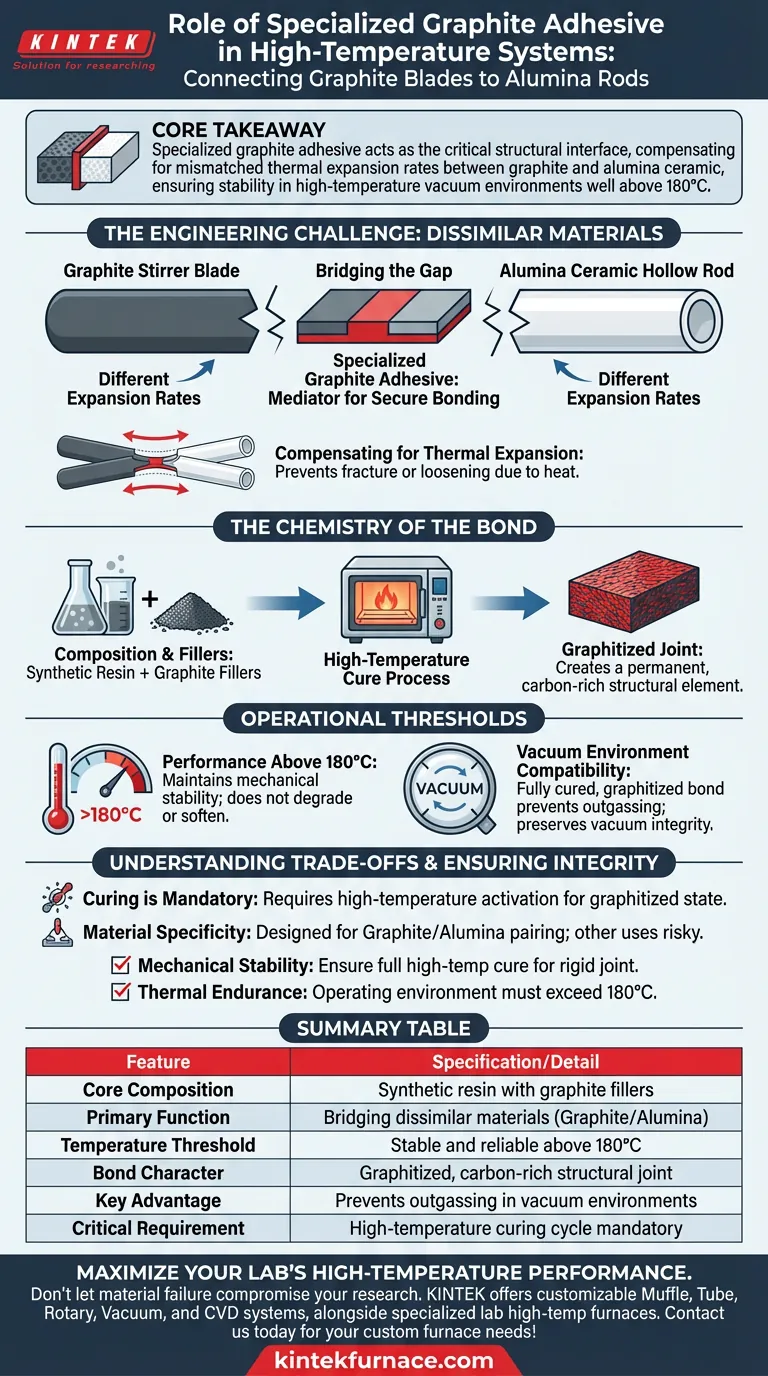
Specialized graphite adhesive acts as the critical structural interface connecting graphite stirrer blades to alumina ceramic hollow rods. Composed of synthetic resin and graphite fillers, this material creates a robust, graphitized joint designed specifically to survive conditions where mechanical fasteners or standard epoxies would fail.
Core Takeaway Connecting dissimilar materials in extreme environments creates significant mechanical stress. Specialized graphite adhesive solves this by compensating for mismatched thermal expansion rates, ensuring the assembly remains stable in high-temperature vacuum environments well above 180°C.

The Engineering Challenge: Dissimilar Materials
Bridging the Gap Between Graphite and Alumina
In high-temperature systems, you are often tasked with joining a graphite blade to an alumina ceramic rod.
These two materials have distinct physical properties.
The adhesive serves as a mediator, bonding the transmission rod to the blade securely despite their material differences.
Compensating for Thermal Expansion
The primary function of this adhesive is to manage thermal expansion.
Graphite and alumina expand at different rates when heated.
Without a flexible or compensating interface, this mismatch would generate stress that causes the joint to fracture or loosen.
The Chemistry of the Bond
Composition and Fillers
The adhesive relies on a specific formulation of synthetic resin and graphite fillers.
This mixture provides the necessary viscosity for application and the chemical backbone for high-heat resistance.
The graphite fillers ensure the bond shares thermal characteristics similar to the blade itself.
The Graphitization Process
This is not a standard room-temperature glue; it requires a high-temperature cure.
During this curing process, the resin creates a strong, graphitized joint.
This transformation turns the adhesive layer into a permanent, carbon-rich structural element.
Operational Thresholds
Performance Above 180°C
Standard adhesives typically degrade, soften, or outgas when temperatures rise.
This specialized adhesive is engineered to maintain mechanical stability at temperatures above 180°C.
It ensures the stirrer blade does not detach during intense heating cycles.
Vacuum Environment Compatibility
The reference highlights the adhesive's role in final high-temperature vacuum environments.
In a vacuum, volatile materials outgas and contaminate the system.
The fully cured, graphitized nature of this bond prevents outgassing, preserving the vacuum's integrity.
Understanding the Trade-offs
The Necessity of Curing
The adhesive does not function instantly; it depends entirely on the curing cycle.
You must heat the assembly to high temperatures to activate the resin and achieve the graphitized state.
Failure to execute this process correctly will result in a weak bond that lacks the necessary thermal properties.
Material Specificity
This solution is highly specialized for graphite and alumina interfaces.
It is designed to compensate for specific expansion coefficients.
Using this adhesive for materials outside this specific thermal pairing may result in unpredictable mechanical failure.
Ensuring Connection Integrity
To ensure your high-temperature assembly succeeds, apply the following principles:
- If your primary focus is Mechanical Stability: Ensure the adhesive undergoes the full high-temperature cure cycle to form a rigid, graphitized joint.
- If your primary focus is Thermal Endurance: Verify that your operating environment exceeds 180°C, where this adhesive outperforms standard bonding agents.
By utilizing the correct curing process, you transform a multi-material assembly into a single, thermally unified component.
Summary Table:
| Feature | Specification/Detail |
|---|---|
| Core Composition | Synthetic resin with graphite fillers |
| Primary Function | Bridging dissimilar materials (Graphite/Alumina) |
| Temperature Threshold | Stable and reliable above 180°C |
| Bond Character | Graphitized, carbon-rich structural joint |
| Key Advantage | Prevents outgassing in vacuum environments |
| Critical Requirement | High-temperature curing cycle mandatory |
Maximize Your Lab’s High-Temperature Performance
Don't let material failure compromise your research. Backed by expert R&D and precision manufacturing, KINTEK offers a comprehensive range of Muffle, Tube, Rotary, Vacuum, and CVD systems, alongside specialized lab high-temp furnaces—all fully customizable to meet your unique materials science challenges.
Whether you are bonding dissimilar materials or require high-vacuum integrity, our engineering team is ready to provide the solution. Contact us today to discuss your custom furnace needs!
Visual Guide
References
- Younès Belrhiti, Amal Chabli. Mechanical stirring: Novel engineering approach for in situ spectroscopic analysis of melt at high temperature. DOI: 10.1016/j.heliyon.2024.e25626
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Molybdenum Disilicide MoSi2 Thermal Heating Elements for Electric Furnace
- 1400℃ Muffle Oven Furnace for Laboratory
- Silicon Carbide SiC Thermal Heating Elements for Electric Furnace
- Vacuum Hot Press Furnace Machine Heated Vacuum Press Tube Furnace
- HFCVD Machine System Equipment for Drawing Die Nano Diamond Coating
People Also Ask
- What ceramic materials are commonly used for heating elements? Discover the Best for Your High-Temp Needs
- What is the temperature range where MoSi2 heating elements should not be used for long periods? Avoid 400-700°C to Prevent Failure
- What types of molybdenum disilicide heating elements are available? Choose the Right Element for Your High-Temp Needs
- What are the primary applications of MoSi2 heating elements in research? Achieve Reliable High-Temp Control for Material Synthesis
- What are the advantages of using molybdenum-disilicide heating elements for aluminum alloy processing? (Rapid Heating Guide)











