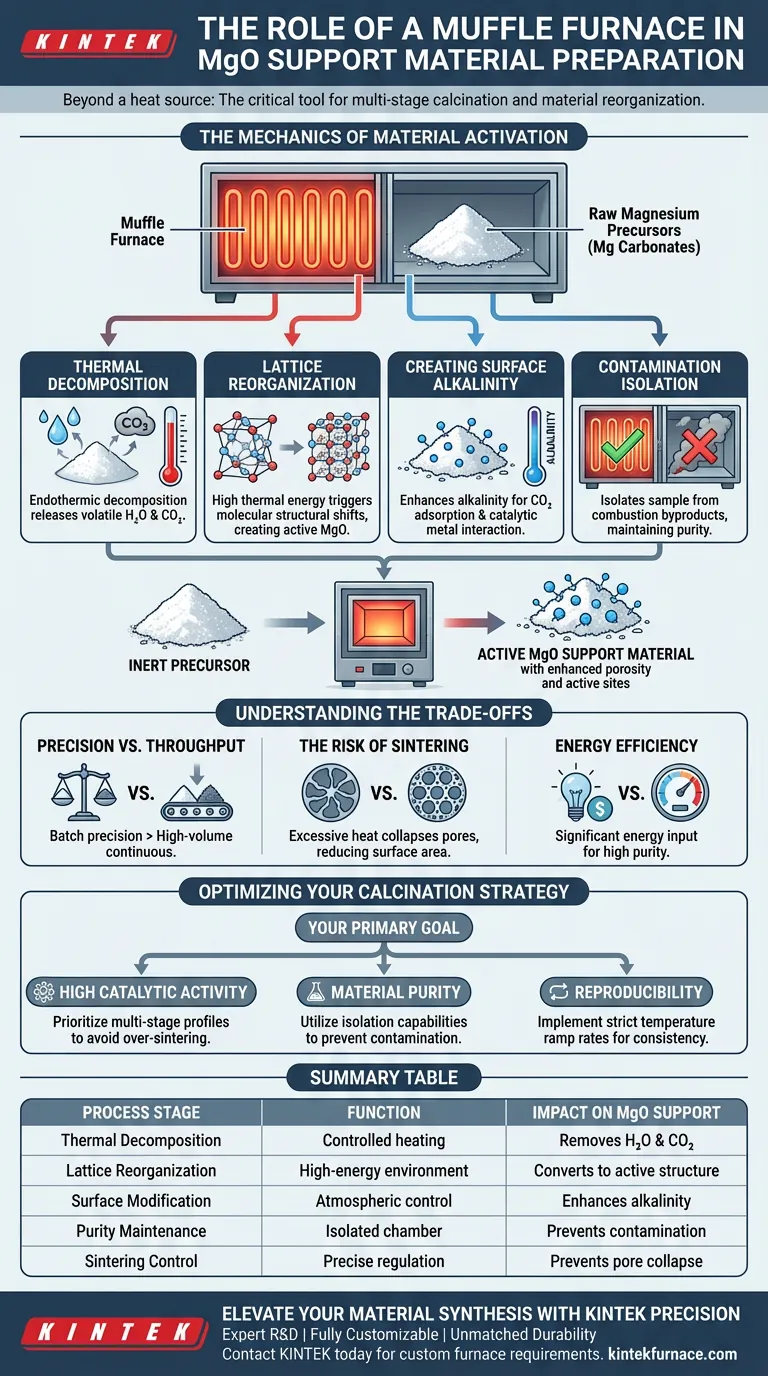
A muffle furnace serves as the critical thermal processing environment required to transform raw magnesium precursors into high-performance support materials. It provides a precise, high-temperature setting that facilitates the thermal decomposition of magnesium carbonates, a necessary step for creating active Magnesium Oxide (MgO).
Core Takeaway The muffle furnace is not merely a heat source; it is the tool that drives multi-stage calcination to reorganize the material's crystal lattice. This process strips away water and carbon dioxide, leaving behind a highly alkaline, active MgO structure optimized for metal loading and catalytic performance.
The Mechanics of Material Activation
Thermal Decomposition
The primary function of the furnace is to drive the endothermic decomposition of magnesium carbonates. By maintaining specific high temperatures, the furnace forces the release of volatile components.
This effectively removes chemically bound water and carbon dioxide from the raw material structure.
Lattice Reorganization
Beyond simple drying, the muffle furnace induces physical changes at the molecular level. The high thermal energy triggers a reorganization of the crystal lattice.
This structural shift is what converts inert precursors into "active" magnesium oxide. Without this precise thermal history, the material would lack the necessary surface properties to act as a support.
Creating Surface Alkalinity
The controlled environment allows for the development of specific chemical traits. The process directly enhances the alkalinity of the final MgO product.
High alkalinity is crucial for the material's ability to adsorb carbon dioxide and interact effectively with catalytic metals during later synthesis stages.
Contamination Isolation
A distinct advantage of the muffle furnace is its ability to isolate the sample from fuel combustion byproducts. The heating elements are separated from the reaction chamber.
This ensures that the chemical purity of the MgO is maintained, preventing sulfur or carbon contamination from the heat source itself.
Understanding the Trade-offs
Precision vs. Throughput
Muffle furnaces excel at batch processing where temperature precision is paramount. However, they are generally less suited for continuous, high-volume industrial production compared to rotary kilns.
The Risk of Sintering
While high heat is necessary for activation, excessive temperature or duration can lead to over-sintering. This collapses the pore structure, drastically reducing surface area and catalytic activity.
Energy Efficiency
Reaching and maintaining temperatures above 500°C–1000°C requires significant energy input. Using a muffle furnace for support preparation requires balancing the cost of energy against the value of the resulting material purity.
Optimizing Your Calcination Strategy
To maximize the quality of your MgO support materials, align your furnace parameters with your specific chemical goals.
- If your primary focus is high catalytic activity: Prioritize a multi-stage calcination profile to ensure complete lattice reorganization without over-sintering the pores.
- If your primary focus is material purity: Utilize the muffle furnace's isolation capabilities to prevent combustion byproducts from interfering with the MgO surface chemistry.
- If your primary focus is reproducibility: Implement strict temperature ramp rates to ensure every batch undergoes the exact same thermal history.
The muffle furnace is the gatekeeper of quality, determining whether your raw precursor becomes a high-performance catalytic platform or merely inert dust.
Summary Table:
| Process Stage | Function of Muffle Furnace | Impact on MgO Support |
|---|---|---|
| Thermal Decomposition | Controlled heating of magnesium carbonates | Removes $H_2O$ and $CO_2$ to form MgO |
| Lattice Reorganization | High-energy thermal environment | Converts precursors into an active crystal structure |
| Surface Modification | Atmospheric control and high temp | Enhances alkalinity for superior metal loading |
| Purity Maintenance | Isolated heating chamber | Prevents contamination from fuel combustion byproducts |
| Sintering Control | Precise temperature regulation | Prevents pore collapse to maintain high surface area |
Elevate Your Material Synthesis with KINTEK Precision
Unlock the full potential of your MgO supports with thermal solutions engineered for excellence. KINTEK provides industry-leading muffle, tube, and vacuum systems designed to give you total control over lattice reorganization and surface chemistry.
Why choose KINTEK for your lab or production facility?
- Expert R&D: Our systems are backed by advanced research to ensure uniform heating and contamination-free processing.
- Fully Customizable: Whether you need a standard muffle furnace or a specialized CVD system, we tailor our technology to your unique research needs.
- Unmatched Durability: Built for high-temp stability, ensuring reproducible results batch after batch.
Ready to optimize your calcination strategy? Contact KINTEK today to discuss your custom furnace requirements and see how our expert manufacturing can advance your catalytic performance.
Visual Guide
References
- Kamonrat Suksumrit, Susanne Lux. Reduced Siderite Ore Combined with Magnesium Oxide as Support Material for Ni-Based Catalysts; An Experimental Study on CO2 Methanation. DOI: 10.3390/catal14030206
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Laboratory Muffle Oven Furnace with Bottom Lifting
- 1800℃ High Temperature Muffle Oven Furnace for Laboratory
- 1700℃ High Temperature Muffle Oven Furnace for Laboratory
- 1400℃ Muffle Oven Furnace for Laboratory
- 1700℃ High Temperature Laboratory Tube Furnace with Alumina Tube
People Also Ask
- Why is high-purity argon protection required during the casting of alloy steel samples? Preserve Sample Integrity
- How does the choice of ceramic molds affect the results when preparing steel sample ingots? Ensure Maximum Sample Purity
- How does a laboratory furnace address the strength-ductility trade-off in UFG titanium? Master Thermal Processing.
- What is the function of vacuum & heating in aluminum degassing? Enhancing Composite Integrity & Density
- What functions does glucose perform in lithium-ion sieve synthesis? Enhance Carbothermal Reduction for LiMnO2 Purity



















