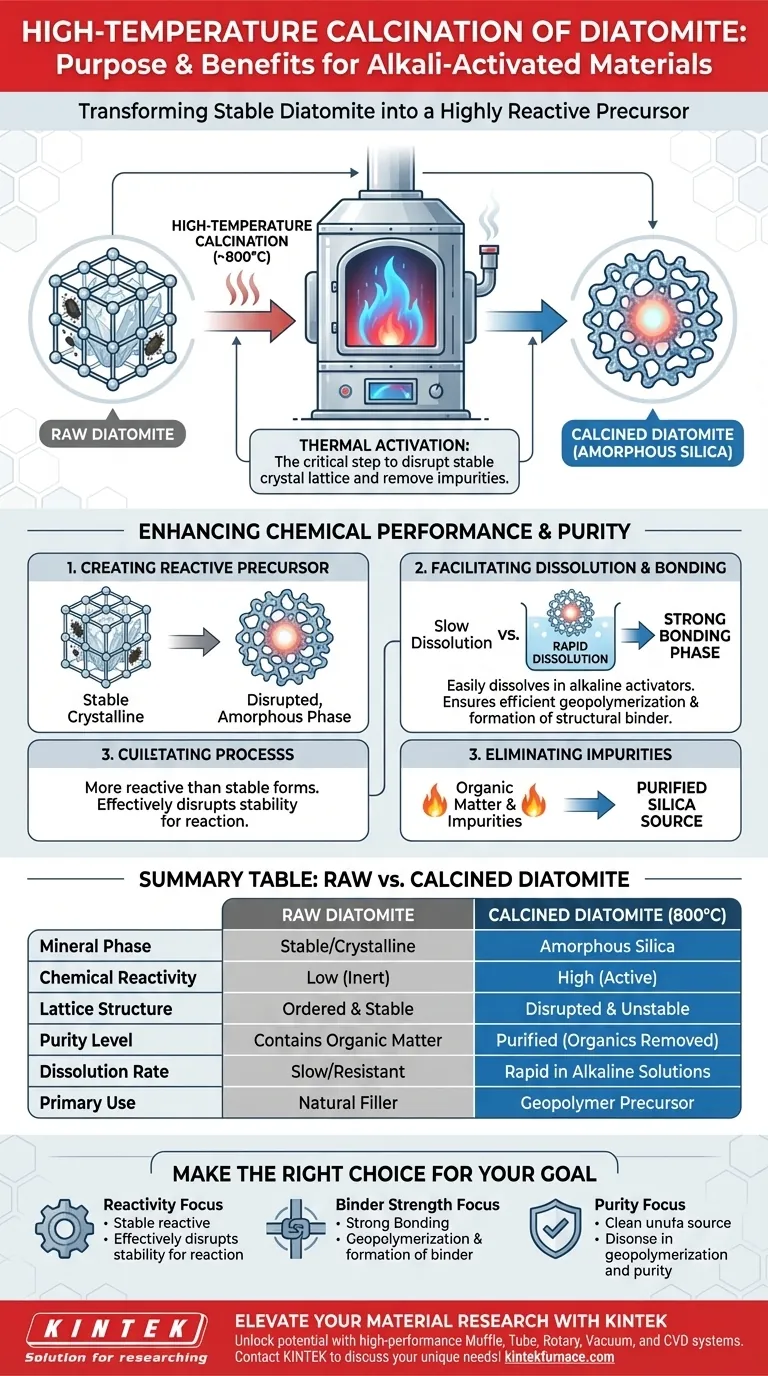
The primary purpose of high-temperature calcination is to transform stable diatomite into a highly reactive precursor for alkali-activated materials. By subjecting the material to temperatures around 800°C, the process converts the natural siliceous components into amorphous silica. This thermal treatment is essential to disrupt the material's stable crystal lattice and remove impurities, rendering it capable of efficient geopolymerization.
Natural diatomite is often too stable to participate effectively in chemical bonding. Calcination acts as a critical activation step, breaking down the material's internal structure to ensure it dissolves readily in alkaline solutions to form a strong binder.

The Mechanism of Thermal Activation
Creating Amorphous Silica
The core objective of calcining diatomite at 800°C is to alter its mineralogical phase.
The heat transforms the siliceous components into amorphous silica (SiO2). This amorphous state is significantly more reactive than the crystalline or stable forms found in raw diatomite.
Disrupting the Lattice Structure
Natural diatomite possesses a stable lattice structure that resists chemical attack.
High-temperature processing effectively disrupts this stability. By breaking the lattice, the material becomes thermodynamically unstable and primed for chemical reaction.
Enhancing Chemical Performance
Facilitating Dissolution
For alkali-activated materials to form, the solid precursor must first dissolve in the activator solution.
The structural disruption caused by calcination allows the diatomite to dissolve more easily in alkaline activators. This rapid dissolution is the trigger for the subsequent geopolymerization process.
Strengthening the Bonding Phase
The ultimate goal of this preparation is the formation of a structural binder.
Because the calcined material dissolves and reacts more efficiently, it participates more fully in geopolymerization reactions. This results in the enhanced formation of the bonding phase, which dictates the final strength of the material.
Optimizing Material Purity
Eliminating Organic Impurities
Raw diatomite often contains organic matter that can interfere with chemical reactions or weaken the final product.
The thermal process acts as a purification step. The high heat effectively burns off these organic impurities, leaving behind a cleaner silica source.
Understanding the Process Requirements
Equipment and Energy Implications
Achieving the necessary phase change requires specific, high-energy environments.
The process is typically conducted in high-temperature muffle or tube furnaces. While this ensures the conversion to reactive silica, it adds a processing layer that requires controlled thermal management compared to using raw materials.
Making the Right Choice for Your Goal
To maximize the effectiveness of your alkali-activated materials, you must view calcination as a mandatory activation step rather than optional processing.
- If your primary focus is Reactivity: Prioritize calcination at 800°C to ensure the maximum conversion of stable silica into reactive amorphous silica.
- If your primary focus is Binder Strength: Use calcined diatomite to ensure full dissolution in the activator, which is required to form a dense, robust bonding phase.
- If your primary focus is Material Purity: Rely on this thermal treatment to eliminate organic contaminants that could compromise the integrity of the geopolymer matrix.
High-temperature calcination is the bridge that turns raw, inert earth into a chemically active construction material.
Summary Table:
| Feature | Raw Diatomite | Calcined Diatomite (800°C) |
|---|---|---|
| Mineral Phase | Stable/Crystalline | Amorphous Silica |
| Chemical Reactivity | Low (Inert) | High (Active) |
| Lattice Structure | Ordered & Stable | Disrupted & Unstable |
| Purity Level | Contains Organic Matter | Purified (Organics Removed) |
| Dissolution Rate | Slow/Resistant | Rapid in Alkaline Solutions |
| Primary Use | Natural Filler | Geopolymer Precursor |
Elevate Your Material Research with KINTEK
Unlock the full potential of your alkali-activated materials with precision thermal processing. Backed by expert R&D and manufacturing, KINTEK offers high-performance Muffle, Tube, Rotary, Vacuum, and CVD systems, all fully customizable to meet your specific calcination requirements. Whether you are activating diatomite or developing advanced geopolymers, our lab high-temp furnaces provide the uniform heating and precise control needed for superior results.
Ready to optimize your calcination process? Contact KINTEK today to discuss your unique needs with our technical team!
Visual Guide
References
- Darius Žūrinskas, Danutė Vaičiukynienė. Mechanical and Microstructural Properties of Alkali-Activated Biomass Fly Ash and Diatomite Blends. DOI: 10.3390/ma18163807
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- High Temperature Muffle Oven Furnace for Laboratory Debinding and Pre Sintering
- 1200℃ Muffle Oven Furnace for Laboratory
- 1700℃ High Temperature Muffle Oven Furnace for Laboratory
- 1800℃ High Temperature Muffle Oven Furnace for Laboratory
- 1400℃ High Temperature Laboratory Tube Furnace with Alumina Tube
People Also Ask
- What role does a high-temperature muffle furnace play in STFO synthesis? Achieve Pure Perovskite Results
- How is a muffle furnace utilized during the high-temperature annealing of forged TiAl-SiC composites?
- What role does a high-temperature muffle furnace play in TiO2 and PEN cross-linking? Unlock High-Performance Hybrids
- How does precise temperature control affect MoS2/rGO hybrids? Mastering Nanowall Morphology
- What is the primary function of a high-temperature muffle furnace for cerium dioxide precursors? Expert Calcination Tips



















