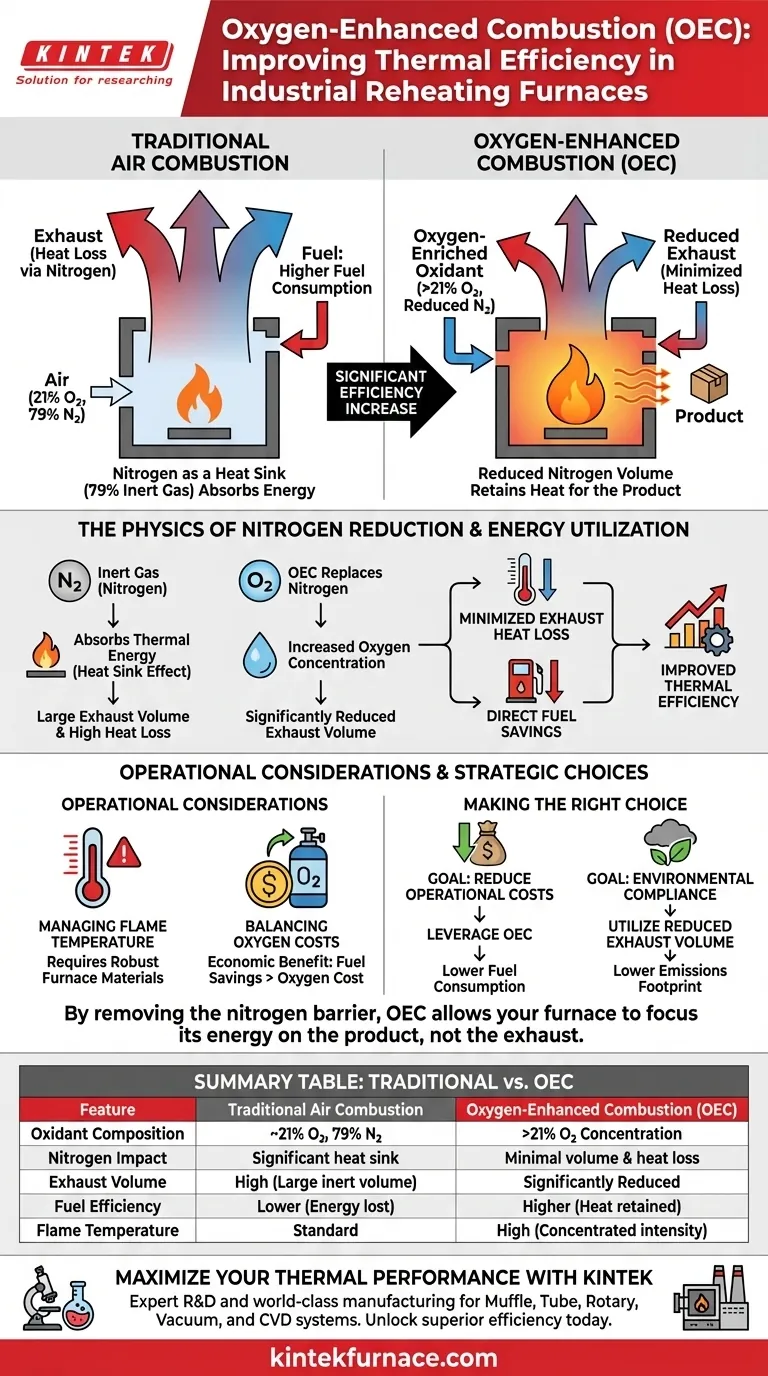
Oxygen-Enhanced Combustion (OEC) fundamentally transforms thermal efficiency by increasing the concentration of oxygen in the oxidant stream above the standard 21% found in atmospheric air. This process systematically eliminates the volume of nitrogen—an inert gas that inhibits efficiency—resulting in significantly reduced exhaust gas volumes, lower heat losses, and reduced fuel consumption.
By increasing the oxidant’s oxygen level above 21%, OEC drastically minimizes nitrogen volume. This directly lowers exhaust heat losses and fuel consumption, resulting in a significantly more efficient thermal process compared to traditional air combustion.

The Physics of Nitrogen Reduction
The Role of Inert Gases
In traditional air combustion, nearly 79% of the air intake is nitrogen. This nitrogen does not participate in the combustion reaction; it is chemically inert in this context.
The Heat Sink Effect
Despite being inert, nitrogen absorbs a massive amount of thermal energy. It acts as a "heat sink," stealing energy from the flame that should be directed toward the product being heated in the furnace.
Reducing Exhaust Volume
OEC technology replaces this nitrogen with oxygen. Because there is less inert gas flowing through the system, the overall volume of exhaust gas produced is significantly reduced.
Enhancing Energy Utilization
Minimizing Exhaust Heat Loss
The primary driver of efficiency in OEC is the reduction of "sensible heat" lost through the stack. When you reduce the volume of hot exhaust gas leaving the furnace, you retain more heat inside the chamber.
Direct Fuel Savings
Because the system no longer wastes energy heating up nitrogen that is simply vented into the atmosphere, the fuel requirement drops. You consume less fuel to achieve the same target temperature for the reheating process.
Improved Thermal Efficiency
The combination of lower fuel input and higher heat retention leads to a sharp increase in overall thermal efficiency. The energy is utilized for the process load rather than heating the atmosphere.
Understanding the Operational Considerations
Managing Flame Temperature
While OEC improves efficiency, the removal of nitrogen (which acts as a thermal ballast) can lead to higher flame temperatures. Operators must ensure furnace materials and refractories can withstand this increased intensity.
Balancing Oxygen Costs
The efficiency gains from fuel savings must be weighed against the cost of generating or purchasing pure oxygen. The economic benefit is realized when the fuel savings outweigh the cost of oxygen supply.
Making the Right Choice for Your Goal
Implementing Oxygen-Enhanced Combustion is a strategic decision that depends on your specific operational bottlenecks.
- If your primary focus is Reducing Operational Costs: Leverage OEC to lower fuel consumption by eliminating the energy wasted on heating inert nitrogen.
- If your primary focus is Environmental Compliance: Utilize the reduction in total exhaust gas volume to lower the overall emissions footprint of your facility.
By removing the nitrogen barrier, OEC allows your furnace to focus its energy on the product, not the exhaust.
Summary Table:
| Feature | Traditional Air Combustion | Oxygen-Enhanced Combustion (OEC) |
|---|---|---|
| Oxidant Composition | ~21% Oxygen, 79% Nitrogen | >21% Oxygen Concentration |
| Nitrogen Impact | Acts as a significant heat sink | Minimal nitrogen volume and heat loss |
| Exhaust Volume | High (large volume of inert gas) | Significantly Reduced |
| Fuel Efficiency | Lower (energy lost to stack) | Higher (more heat stays in furnace) |
| Flame Temperature | Standard | High (Concentrated intensity) |
Maximize Your Thermal Performance with KINTEK
Ready to transform your laboratory or industrial heating processes? Backed by expert R&D and world-class manufacturing, KINTEK provides high-performance Muffle, Tube, Rotary, Vacuum, and CVD systems designed for precision and efficiency. Whether you need to optimize energy utilization or require a fully customizable high-temp furnace for unique research needs, our team is here to help.
Unlock superior efficiency today—Contact KINTEK for a tailored solution!
Visual Guide
References
- Minsheng Zhao, Xianzhong Hu. Study on Flow and Heat Transfer Characteristics of Reheating Furnaces Under Oxygen-Enriched Conditions. DOI: 10.3390/pr13082454
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Laboratory Muffle Oven Furnace with Bottom Lifting
- High Temperature Muffle Oven Furnace for Laboratory Debinding and Pre Sintering
- 1400℃ Muffle Oven Furnace for Laboratory
- 1700℃ High Temperature Muffle Oven Furnace for Laboratory
- 1700℃ High Temperature Laboratory Tube Furnace with Alumina Tube
People Also Ask
- What is the purpose of bottom-entry argon injection? Enhance Lithium-ion Battery Safety & Purge Efficiency
- How does a vacuum system integrated with mechanical and diffusion pumps enhance lead carbonate decomposition? Guide
- How does a laboratory furnace address the strength-ductility trade-off in UFG titanium? Master Thermal Processing.
- What functions does glucose perform in lithium-ion sieve synthesis? Enhance Carbothermal Reduction for LiMnO2 Purity
- Why is high-purity argon protection required during the casting of alloy steel samples? Preserve Sample Integrity



















