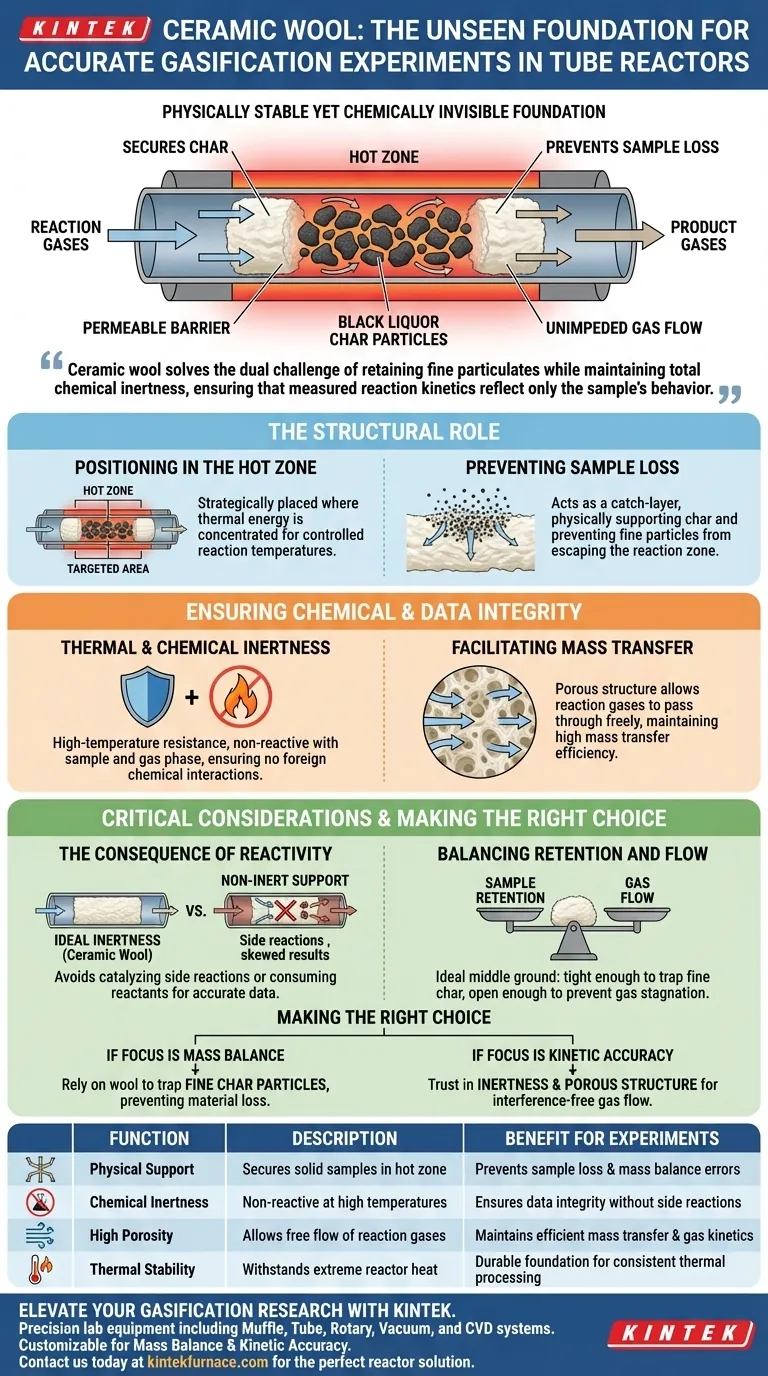
Ceramic wool acts as a physically stable yet chemically invisible foundation for solid samples within the high-temperature environment of a tube reactor. It creates a permeable barrier in the hot zone that secures black liquor char particles, preventing them from falling out of the reaction zone without impeding the flow of essential gases.
Ceramic wool solves the dual challenge of retaining fine particulates while maintaining total chemical inertness, ensuring that measured reaction kinetics reflect only the sample's behavior and not the support material.

The Structural Role of Ceramic Wool
Positioning in the Hot Zone
In gasification experiments, the reaction must occur at specific, controlled temperatures. Ceramic wool is strategically placed directly in the hot zone of the tube reactor.
This positioning ensures that the black liquor char samples are held precisely where the thermal energy is concentrated.
Preventing Sample Loss
One of the primary mechanical functions of ceramic wool is to act as a catch-layer. It physically supports the char samples and prevents fine particles from falling through the reactor tube.
Without this physical barrier, smaller particulates could escape the reaction zone, leading to incomplete data or mass balance errors.
Ensuring Chemical and Data Integrity
Thermal and Chemical Inertness
To obtain accurate gasification measurements, the support material must not become part of the experiment. Ceramic wool possesses excellent high-temperature resistance, allowing it to withstand the reactor's heat without degrading.
Furthermore, it is chemically inert. It remains non-reactive with both the solid char sample and the surrounding gas phase, ensuring no foreign chemical interactions skew the results.
Facilitating Mass Transfer
While the wool must hold the solid sample, it must not block the gas flow. Ceramic wool features a highly porous structure that allows reaction gases to pass through freely.
This porosity maintains high mass transfer efficiency, which is critical for ensuring the gasification process proceeds uninhibited.
Critical Considerations for Experimental Integrity
The Consequence of Reactivity
A common pitfall in reactor design is selecting a support material that interacts with the sample. If the support layer is not fully inert, it may catalyze side reactions or consume reactants.
Ceramic wool is specifically utilized to avoid this variable, ensuring that changes in the gas phase are attributed solely to the char gasification.
Balancing Retention and Flow
The trade-off in support material selection lies between holding the sample and allowing gas flow. A material that is too dense might hold fines well but choke the reactor.
Ceramic wool serves as the ideal middle ground, offering a matrix tight enough to trap fine char but open enough to prevent back-pressure or gas stagnation.
Making the Right Choice for Your Experiment
To ensure your gasification data is reliable, apply the specific properties of ceramic wool to your experimental goals:
- If your primary focus is Mass Balance: Rely on the ceramic wool to trap fine char particles, preventing material loss that would ruin gravimetric analysis.
- If your primary focus is Kinetic Accuracy: Trust in the chemical inertness and porous structure of the wool to allow gases to flow without interference.
By utilizing ceramic wool, you secure the physical sample without compromising the chemical validity of the reaction environment.
Summary Table:
| Function | Description | Benefit for Experiments |
|---|---|---|
| Physical Support | Secures solid samples/char in the hot zone | Prevents sample loss and mass balance errors |
| Chemical Inertness | Non-reactive at high temperatures | Ensures data integrity without side reactions |
| High Porosity | Allows free flow of reaction gases | Maintains efficient mass transfer and gas kinetics |
| Thermal Stability | Withstands extreme reactor heat | Durable foundation for consistent thermal processing |
Elevate Your Gasification Research with KINTEK
Precision in the lab starts with the right equipment. Backed by expert R&D and manufacturing, KINTEK offers high-performance Muffle, Tube, Rotary, Vacuum, and CVD systems, all fully customizable to meet your unique experimental needs. Whether you are optimizing mass balance or ensuring kinetic accuracy, our lab high-temp furnaces provide the thermal stability your research demands.
Ready to upgrade your thermal processing? Contact us today to consult with our experts and find the perfect reactor solution for your laboratory.
Visual Guide
References
- F. Bueno, José Luis Sánchez. CO₂ Gasification of Black Liquor Char under isothermal and dynamic conditions. DOI: 10.26754/jji-i3a.202512008
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Laboratory Quartz Tube Furnace RTP Heating Tubular Furnace
- Custom Made Versatile CVD Tube Furnace Chemical Vapor Deposition CVD Equipment Machine
- Slide PECVD Tube Furnace with Liquid Gasifier PECVD Machine
- Inclined Rotary Plasma Enhanced Chemical Deposition PECVD Tube Furnace Machine
- Inclined Rotary Plasma Enhanced Chemical Deposition PECVD Tube Furnace Machine
People Also Ask
- What role does a laboratory tube furnace system play in the catalytic pyrolysis of LLDPE? Enhancing Yield and Precision
- What physical conditions does a laboratory Tube Furnace provide for SOEC? Precision Heat for Solid Oxide Characterization
- Why is control of heating rate and gas flow in a lab tube furnace critical for EM wave absorption materials?
- What is the function of a laboratory tube furnace in BiFeO3 nanopowder production? Master the Calcination Stage
- What role does a laboratory tube furnace play in the heat treatment of NiTiCu alloys? Optimize Shape Memory Properties



















