The Allure of the Red Line
In science and engineering, we are instinctively drawn to the upper limit of a specification sheet. The highest frequency, the fastest speed, the greatest pressure. When it comes to a tube furnace, that number is maximum temperature. It’s easy to believe that a furnace capable of 1800°C is inherently "better" than one that tops out at 1200°C.
This is a cognitive trap.
Imagine a materials scientist trying to synthesize a novel perovskite. The protocol calls for 1100°C. They run the experiment in a top-of-the-line 1800°C furnace, yet the sample fails—the crystal structure is impure. The problem wasn't the furnace's peak capability; it was a subtle inconsistency in the heat profile, or an imperceptible atmospheric leak.
The right furnace isn't the one with the highest number. It's the one that creates the most precise and repeatable environment for your specific process.
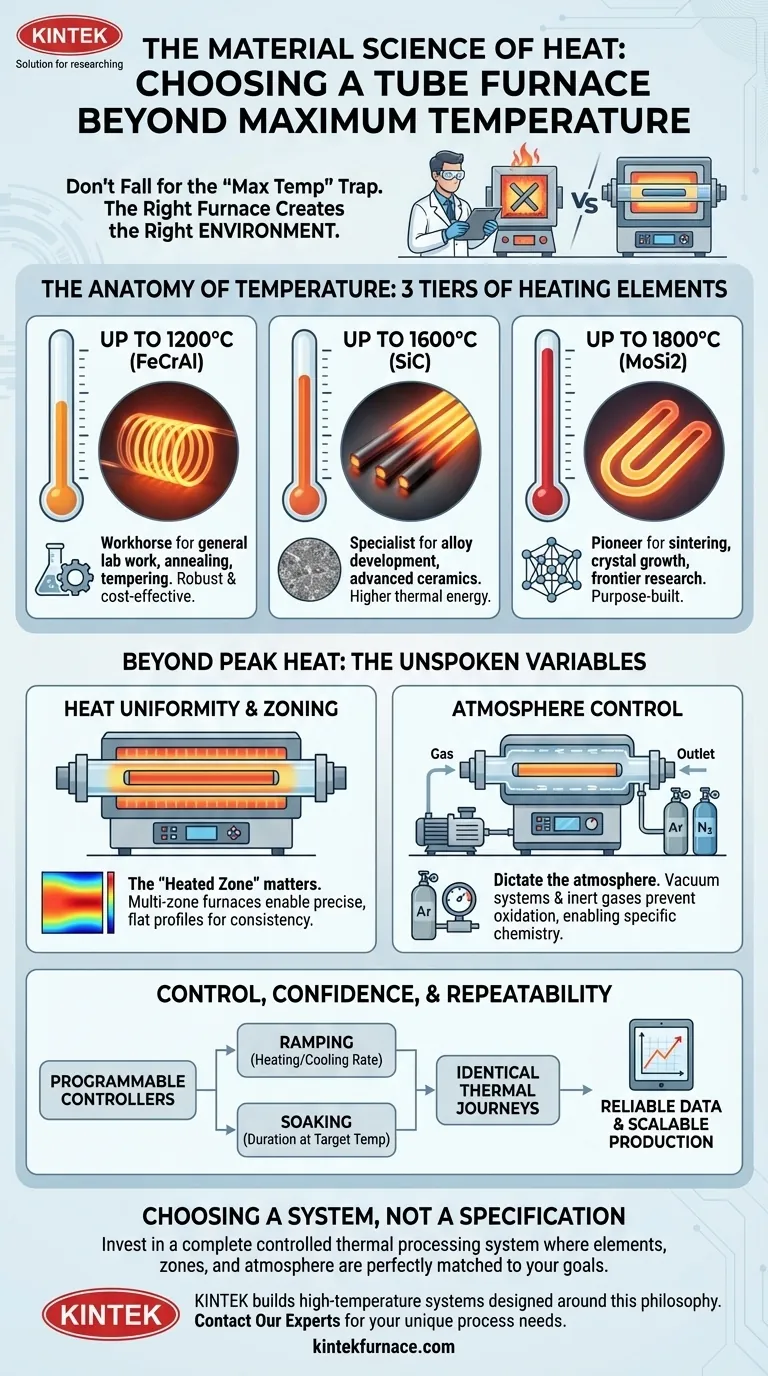
The Anatomy of Temperature
A furnace's maximum temperature is dictated by the fundamental physics of its heating elements. Different materials survive and perform optimally in different thermal regimes. This creates distinct tiers of capability.
The Workhorse: Up to 1200°C (FeCrAl)
Furnaces in this class often use iron-chrome-aluminum alloy (Kanthal) elements. They are robust, reliable, and cost-effective. These are the daily drivers of the lab, perfect for a vast range of applications like annealing, tempering, and general materials synthesis where extreme heat is unnecessary.
The Specialist: Up to 1600°C (SiC)
To push past 1200°C, more exotic materials are needed. Silicon Carbide (SiC) elements enter the scene, enabling higher thermal energy for more demanding tasks. This is the realm of certain ceramic processing, alloy development, and advanced materials research.
The Pioneer: Up to 1800°C (MoSi2)
Reaching the frontier requires the most specialized elements. Molybdenum Disilicide (MoSi2) can operate continuously at blistering temperatures. These furnaces are not for general use; they are purpose-built for advanced sintering of technical ceramics, growing specialized crystals, and testing materials at the edge of their operational limits.
Beyond Peak Heat: The Unspoken Variables
A successful thermal process is a symphony of controlled variables. Temperature is just the conductor; uniformity and atmosphere are the lead instruments.
The Geography of Heat: Uniformity and Zoning
Simply reaching 1800°C at one point is meaningless if your sample experiences a 50°C gradient across its length. The critical metric is the heated zone—the region where temperature is stable and uniform.
For highly sensitive processes, multi-zone furnaces offer granular control. By using several independent heating elements and controllers, you can engineer a perfectly flat temperature profile or even create a deliberate, precise gradient. This transforms the furnace from a simple heater into a precision instrument.
Crafting the Void: Atmosphere Control
Many reactions cannot tolerate the chaotic, oxidative nature of ambient air. True process control often means dictating the atmosphere itself.
Sealed flanges and vacuum systems allow you to remove the atmosphere entirely, down to levels of 10⁻⁵ torr. From there, you can introduce a controlled blanket of inert gas like argon or nitrogen. This creates a pristine stage, preventing unwanted reactions and enabling the specific chemistry you designed.
Control, Confidence, and Repeatability
The ultimate goal of any scientific process is a repeatable result. This is where the human need for confidence meets engineering precision.
Modern programmable controllers allow you to dictate the entire thermal journey:
- Ramping: The rate of heating and cooling.
- Soaking: The duration spent at a target temperature.
This level of control removes guesswork. It ensures that the process run on Tuesday is identical to the one run three months later, forming the bedrock of reliable data and scalable production.
Choosing a System, Not a Specification
The choice of a furnace is a trade-off between ambition and practicality. The materials science that enables 1800°C operation—from the MoSi2 elements to the high-purity alumina insulation—comes at a significant cost and requires stricter operating protocols.
Your decision should be based on the demands of your work.
| Temperature Range | Common Heating Element | Best For... |
|---|---|---|
| Up to 1200°C | Iron-Chrome-Aluminum (Kanthal) | General lab work, annealing, tempering, calibration |
| Up to 1600°C | Silicon Carbide (SiC) | Alloy development, specific ceramics, advanced materials |
| Up to 1800°C | Molybdenum Disilicide (MoSi2) | High-temp sintering, crystal growth, frontier research |
Ultimately, you are not just buying a furnace; you are investing in a controlled thermal processing system. The right choice is one where the heating elements, temperature zones, and atmosphere management are all perfectly matched to your scientific goals.
As a dedicated R&D and manufacturing expert, KINTEK builds high-temperature systems—from Tube and Muffle Furnaces to advanced CVD platforms—that are designed around this philosophy. We specialize in customizing these complex systems to deliver the precise, repeatable environments your unique work demands. To find the right solution for your process, Contact Our Experts.
Visual Guide
Related Products
- 1700℃ High Temperature Laboratory Tube Furnace with Alumina Tube
- 1400℃ High Temperature Laboratory Tube Furnace with Alumina Tube
- 1200℃ Split Tube Furnace Laboratory Quartz Tube Furnace with Quartz Tube
- Vertical Laboratory Quartz Tube Furnace Tubular Furnace
- High Pressure Laboratory Vacuum Tube Furnace Quartz Tubular Furnace
Related Articles
- Your Furnace Isn't Just a Heater: Why 'Good Enough' Equipment Is Sabotaging Your Advanced Materials Research
- Mastering the Micro-Environment: The Art and Science of the Laboratory Tube Furnace
- The Controlled Universe: Why Tube Furnaces Prioritize Precision Over Scale
- The Physics of Mismatched Tools: Why a Tube Furnace Isn't for Wastewater
- Mastering the Void: How Tube Furnaces Forge the Atoms of Innovation




















