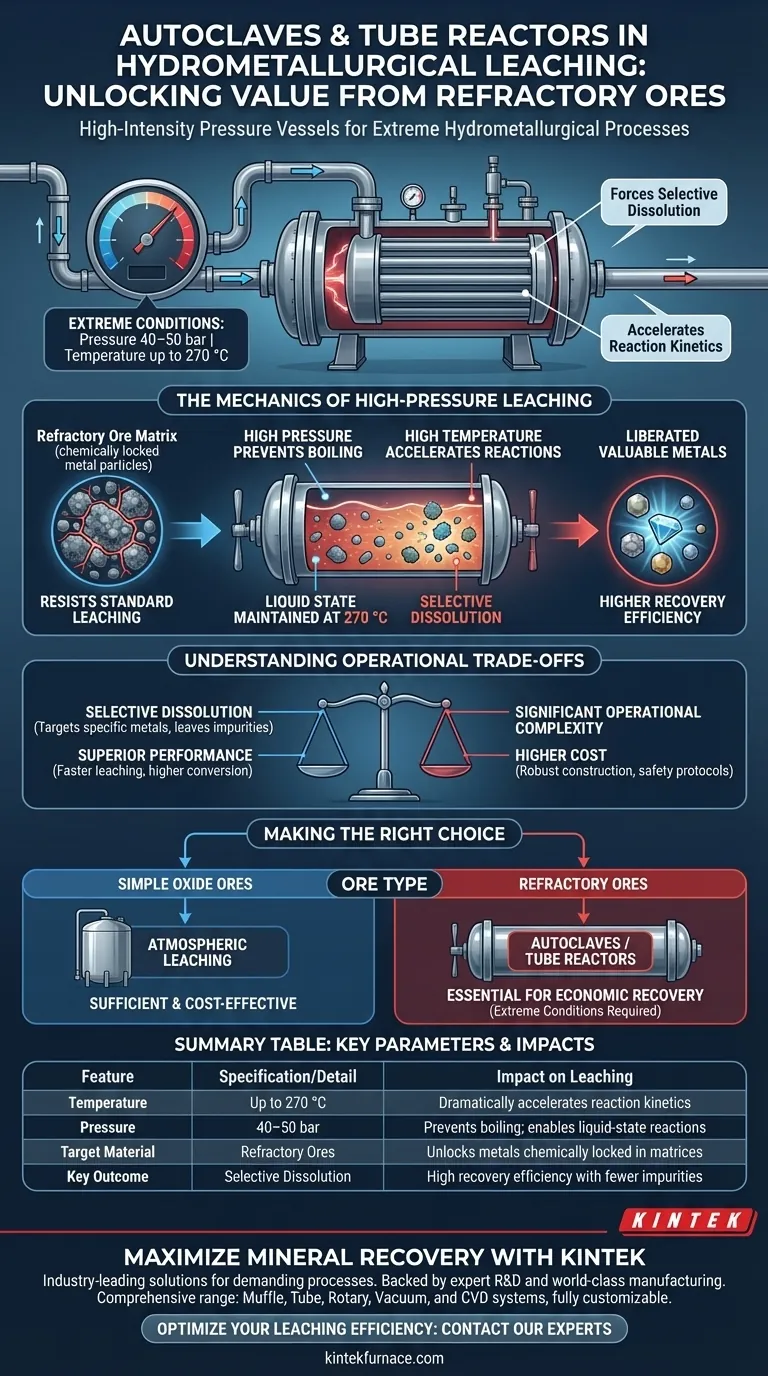
Autoclaves and tube reactors function as high-intensity pressure vessels designed to facilitate hydrometallurgical processes that are impossible under standard atmospheric conditions. Their primary role is to generate and maintain extreme environments—specifically pressures of 40–50 bar and temperatures up to 270 °C—to force the selective dissolution of target metals from complex ores.
By elevating temperature and pressure far beyond atmospheric limits, these reactors dramatically accelerate reaction kinetics, unlocking valuable metals from "refractory" ores that resist standard leaching methods.

The Mechanics of High-Pressure Leaching
Targeting Refractory Ores
Many mineral deposits are classified as refractory, meaning the valuable metal is chemically locked within a matrix that resists standard processing.
Simple atmospheric leaching is often too slow or chemically insufficient to liberate these metals. Autoclaves and tube reactors are engineered specifically to overcome this resistance by subjecting the slurry to aggressive chemical environments.
The Role of Temperature and Pressure
To achieve the necessary chemical conversion, reaction temperatures must reach as high as 270 °C.
At this temperature, liquid solutions would normally boil away instantly. To prevent this, these vessels maintain high pressures of 40–50 bar, keeping the solution in a liquid state and allowing the chemical leaching process to proceed at highly accelerated rates.
Enhancing Chemical Conversion
The combination of high heat and pressure does more than just melt rock; it fundamentally changes the reaction kinetics.
These conditions significantly increase the leaching rate, reducing the time required to extract the metal. Furthermore, they improve the overall conversion efficiency, ensuring a higher percentage of the target metal is recovered from the host rock.
Understanding the Operational Trade-offs
The Cost of Extreme Conditions
While these systems offer superior performance, they introduce significant operational complexity compared to atmospheric tanks.
The requirement to maintain 40–50 bar of pressure necessitates robust, expensive vessel construction and specialized safety protocols.
Selectivity vs. Complexity
The primary advantage of using these reactors is selective dissolution, allowing operators to target specific metals while leaving impurities behind.
However, achieving this selectivity requires precise control over the temperature and pressure profile. Deviations from the optimal 270 °C or pressure drop can lead to stalled reactions or the co-dissolution of unwanted contaminants.
Making the Right Choice for Your Goal
These systems are high-performance tools designed for specific mineralogical challenges.
- If your primary focus is processing simple oxide ores: Avoid these high-pressure systems, as standard atmospheric leaching is sufficient and far more cost-effective.
- If your primary focus is recovering metals from refractory ores: You must utilize autoclaves or tube reactors to achieve the extreme conditions (up to 270 °C) required for economic recovery rates.
High-pressure leaching is the definitive solution for maximizing yield from difficult-to-process mineral deposits.
Summary Table:
| Feature | Specification/Detail | Impact on Leaching |
|---|---|---|
| Temperature | Up to 270 °C | Dramatically accelerates reaction kinetics |
| Pressure | 40–50 bar | Prevents boiling; enables liquid-state reactions |
| Target Material | Refractory Ores | Unlocks metals chemically locked in matrices |
| Key Outcome | Selective Dissolution | High recovery efficiency with fewer impurities |
Maximize Your Mineral Recovery with KINTEK
Don't let refractory ores limit your yield. KINTEK provides industry-leading high-temperature and high-pressure solutions tailored for the most demanding hydrometallurgical processes. Backed by expert R&D and world-class manufacturing, we offer a comprehensive range of Muffle, Tube, Rotary, Vacuum, and CVD systems, all fully customizable to your specific laboratory or industrial needs.
Ready to optimize your leaching efficiency? Contact our technical experts today to design a high-performance system that delivers the precision and durability your project requires.
Visual Guide
References
- Srečko Stopić. Feature Papers in Extractive Metallurgy. DOI: 10.3390/met15070751
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- High Pressure Laboratory Vacuum Tube Furnace Quartz Tubular Furnace
- 1700℃ High Temperature Laboratory Tube Furnace with Alumina Tube
- 1200℃ Split Tube Furnace Laboratory Quartz Tube Furnace with Quartz Tube
- Laboratory Quartz Tube Furnace RTP Heating Tubular Furnace
- Laboratory Vacuum Tilt Rotary Tube Furnace Rotating Tube Furnace
People Also Ask
- What is the function of injecting water in wood thermal modification? Unlock Superior Stability and Hydrophobicity
- What is the function of vacuum & heating in aluminum degassing? Enhancing Composite Integrity & Density
- How does the choice of ceramic molds affect the results when preparing steel sample ingots? Ensure Maximum Sample Purity
- How does a laboratory furnace address the strength-ductility trade-off in UFG titanium? Master Thermal Processing.
- What functions does glucose perform in lithium-ion sieve synthesis? Enhance Carbothermal Reduction for LiMnO2 Purity



















