Vacuum Degassing (VD) drives the formation of spinel inclusions primarily through the erosion of ladle refractories. The process utilizes intense stirring within a low-pressure environment to remove gases and facilitate slag-metal reactions. However, this vigorous physical agitation wears down the ladle lining, introducing magnesium into the slag and creating the chemical conditions necessary for magnesium-aluminum (Mg–Al) spinel inclusions to form.
While Vacuum Degassing is critical for gas removal, the prolonged stirring under vacuum accelerates the degradation of ladle bricks. This erosion enriches the slag with Magnesium Oxide (MgO), which serves as the direct chemical precursor for hard, detrimental spinel inclusions.
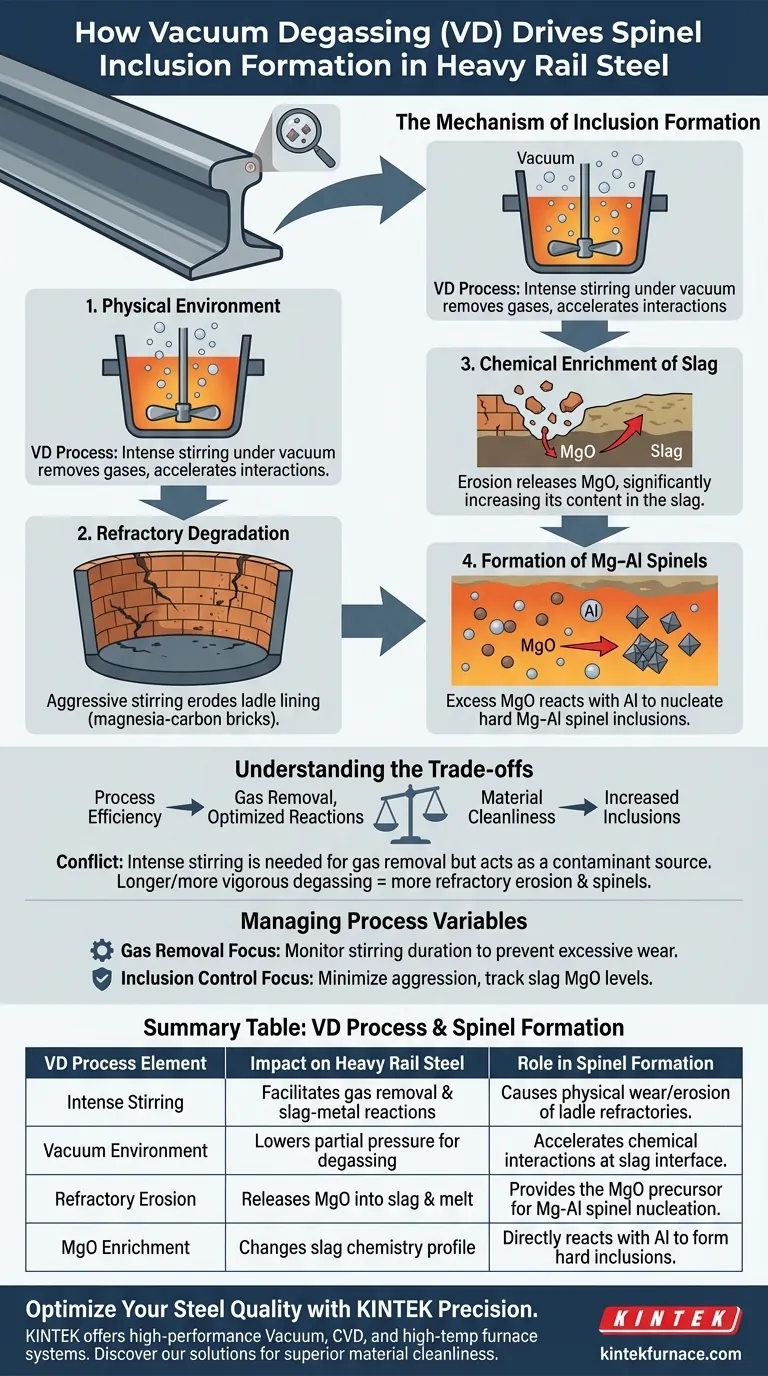
The Mechanism of Inclusion Formation
The Role of Physical Environment
The VD process relies on a vacuum environment combined with prolonged and intense stirring.
This agitation is necessary to ensure the steel melt interacts sufficiently with the slag and to facilitate the removal of unwanted gases.
Refractory Degradation
The aggressive nature of this stirring places immense stress on the ladle refractories.
These linings are typically constructed from magnesia-carbon or high-alumina bricks, which succumb to the physical wear of the process.
Chemical Enrichment of Slag
As the refractories erode, they release their constituent materials into the steelmaking environment.
Specifically, this erosion causes a significant increase in the MgO content within the slag.
Formation of Mg–Al Spinels
The presence of excess MgO in the slag is the tipping point for inclusion formation.
This magnesium reacts with aluminum in the melt, providing the chemical basis for Mg–Al spinel inclusions to nucleate and grow within the heavy rail steel.
Understanding the Trade-offs
Process Efficiency vs. Material Cleanliness
There is an inherent conflict between the operational goals of the VD process and the cleanliness of the final product.
To effectively remove gases and optimize slag-metal reactions, intense stirring is required.
The Cost of Agitation
However, the very energy used to purify the steel of gases acts as a contaminant source regarding inclusions.
Pushing for longer or more vigorous degassing cycles directly correlates with increased refractory erosion and a higher population of spinel inclusions.
Managing Process Variables for Steel Quality
To optimize heavy rail steel production, you must balance the necessity of degassing against the risks of refractory erosion.
- If your primary focus is gas removal: Maintain the necessary stirring intensity but strictly monitor the duration to prevent excessive refractory wear.
- If your primary focus is inclusion control: Minimize the aggression of the stir and closely track MgO levels in the slag to detect early signs of lining degradation.
Understanding that the ladle lining is an active chemical participant in the VD process is key to controlling spinel formation.
Summary Table:
| VD Process Element | Impact on Heavy Rail Steel | Role in Spinel Formation |
|---|---|---|
| Intense Stirring | Facilitates gas removal and slag-metal reactions | Causes physical wear/erosion of ladle refractories |
| Vacuum Environment | Lowers partial pressure for degassing | Accelerates chemical interactions at the slag interface |
| Refractory Erosion | Releases MgO into the slag and melt | Provides the MgO precursor for Mg-Al spinel nucleation |
| MgO Enrichment | Changes slag chemistry profile | Directly reacts with aluminum to form hard inclusions |
Optimize Your Steel Quality with KINTEK Precision
Don't let refractory erosion compromise the integrity of your heavy rail steel. At KINTEK, we understand the delicate balance between efficient vacuum degassing and inclusion control. Backed by expert R&D and manufacturing, we offer high-performance Vacuum, CVD, and laboratory high-temp furnace systems designed to meet the rigorous demands of modern metallurgy.
Whether you need customizable solutions for advanced material research or industrial-grade furnaces for precise thermal processing, our team is ready to help you achieve superior material cleanliness. Contact us today to discover how KINTEK's specialized equipment can enhance your lab's efficiency and production standards.
Visual Guide
References
- Jun Zhu, Jichun Yang. Study on the Causes and Control Measures of Mg–Al Spinel Inclusions in U75V Heavy Rail Steel. DOI: 10.3390/app14051718
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Vacuum Heat Treat Sintering and Brazing Furnace
- Vacuum Heat Treat Sintering Furnace with Pressure for Vacuum Sintering
- Vacuum Heat Treat Sintering Furnace Molybdenum Wire Vacuum Sintering Furnace
- Small Vacuum Heat Treat and Tungsten Wire Sintering Furnace
- Vacuum Hot Press Furnace Machine for Lamination and Heating
People Also Ask
- What are some applications of vacuum brazing? Achieve Strong, Clean Joints in Aerospace and More
- What are the benefits of using vacuum heat treating furnaces for metal alloys? Achieve Superior Metal Properties and Performance
- What is the role of high-precision furnaces in Inconel 718 heat treatment? Master Microstructural Engineering
- What are the advantages of using a vacuum heat treatment furnace? Achieve Superior Material Quality and Control
- What is the temperature of a vacuum furnace brazing? Optimize Your Joint Strength and Cleanliness














