The Alchemist's Modern Dilemma
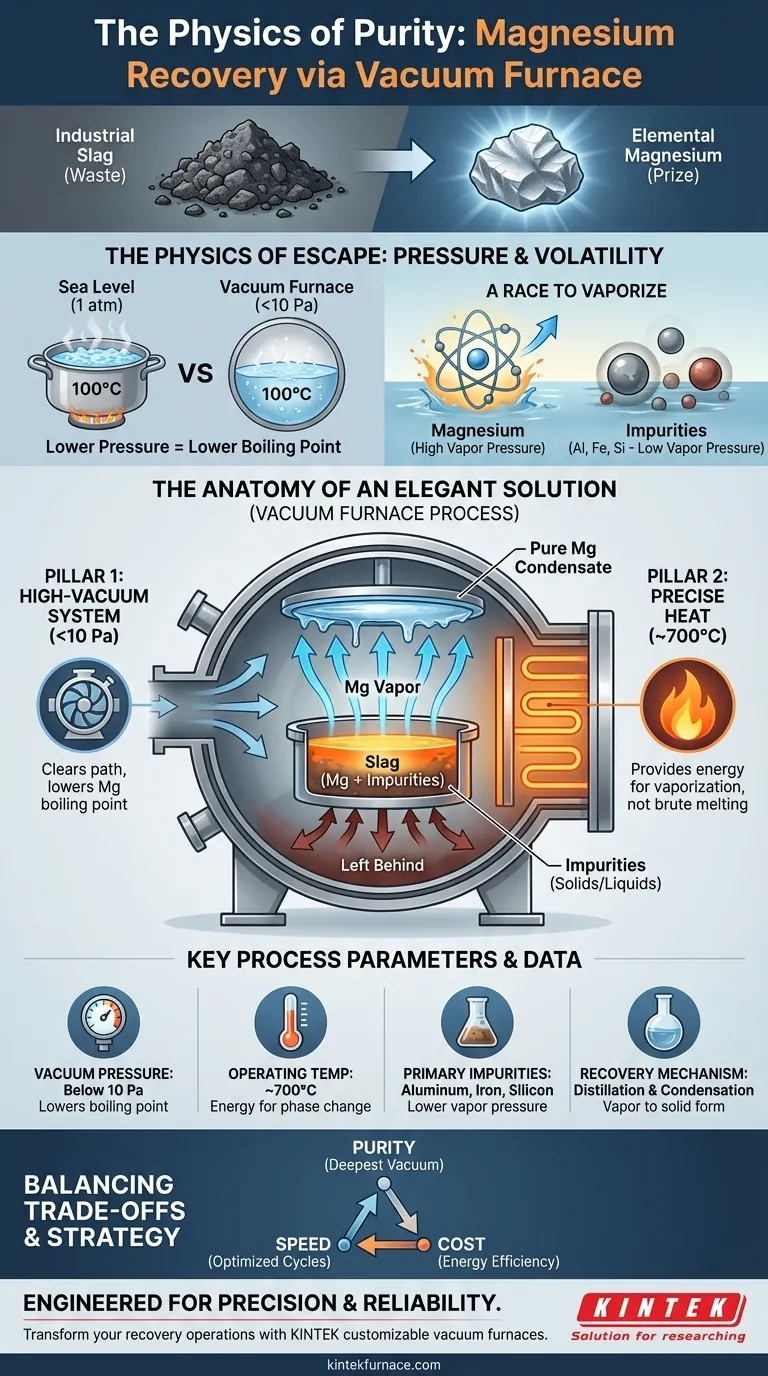
Industrial slag is often seen as waste—a complex, messy byproduct of metallurgical processes. Yet, locked within this dross is a valuable prize: elemental magnesium.
The challenge is not one of chemistry, but of physics and economics. How do you surgically extract a single element from a fused matrix of impurities? Brute force is inefficient. Chemical processes are complex and costly.
The elegant solution lies not in adding more, but in taking something away: air. By creating a vacuum, we can fundamentally alter the rules of nature to our advantage.
The Physics of Escape: Pressure and Volatility
To understand how a vacuum furnace works, you must first forget about simply melting metal. The furnace is a sophisticated physics engine designed to manipulate a substance's state of matter.
At the heart of this process is the inseparable relationship between pressure and boiling point.
Lowering the Bar for Boiling
We all learn that water boils at 100°C. But that's only true at sea-level atmospheric pressure. On a high mountain, where the air is thinner, water boils at a much lower temperature. There's less atmospheric "weight" pushing down on the liquid, making it easier for molecules to escape into a gaseous state.
A vacuum furnace creates an extreme "mountaintop" environment. By pumping out nearly all the air, it reduces the pressure to a tiny fraction of our atmosphere. For magnesium trapped in slag, this changes everything.
A Race to Vaporize
Every element has a natural tendency to become a gas, a property known as its vapor pressure. Think of it as an inherent "desire" to escape its solid or liquid form.
Magnesium has a much higher vapor pressure—a stronger desire to escape—than the common impurities in slag, like aluminum, iron, and silicon.
The vacuum furnace exploits this difference with ruthless efficiency. It creates an environment where magnesium’s escape is not just possible, but inevitable, while its contaminants remain firmly behind.
The Anatomy of an Elegant Solution
The furnace's success depends on the flawless execution of two complementary systems. They are not independent; they are two sides of the same coin, working in concert to achieve a single goal.
Pillar 1: The Power of Emptiness
The high-vacuum system is the star of the show. By reducing the internal pressure to below 10 Pa, it dramatically lowers magnesium's boiling point.
This extreme vacuum doesn't just enable the process; it is the process. It clears a path, removing the atmospheric obstacle and allowing magnesium vapor to travel freely from the molten slag to a cooler condensation surface, where it solidifies into its pure form.
Pillar 2: The Gentle Push of Heat
Heat provides the energy for the escape. The temperature control system must be precise, heating the slag to around 700°C.
This isn't about brute-force melting. The temperature must be just right: hot enough to give the magnesium atoms the energy they need to vaporize in the low-pressure environment, but cool enough to keep the impurities with lower vapor pressures from joining the race. It's a carefully calibrated push, not a shove.
The Unavoidable Trade-offs of Control
Manipulating the laws of physics is powerful, but it comes with inherent challenges. The success of any industrial process is governed by a triangle of competing priorities: Purity, Speed, and Cost.
-
The Cost of Control: Creating a deep vacuum while simultaneously heating a chamber to 700°C is intensely energy-intensive. This is the primary operational cost and a constant factor in economic viability.
-
The Pursuit of Perfection: The entire system relies on an absolutely perfect seal. Even a microscopic leak can compromise the vacuum, raising magnesium's boiling point and destroying the efficiency and purity of the separation.
-
The Rhythm of Production: These furnaces typically operate in batches. The cycle of loading, pumping down, heating, cooling, and unloading creates a deliberate workflow that dictates overall throughput. Speeding up one part of the cycle can compromise another.
Strategy Defines the Machine
How you balance these trade-offs depends entirely on your goal.
- For Maximum Purity: The deepest possible vacuum is your priority. This creates the greatest possible separation between the boiling points of magnesium and its contaminants.
- For Maximum Throughput: The focus shifts to optimizing heating and cooling cycle times, without sacrificing the target vacuum and temperature that guarantee results.
- For Minimum Cost: The goal is to find the "sweet spot"—the perfect equilibrium of vacuum and temperature that achieves the required purity without wasting a single watt of energy.
Ultimately, the vacuum furnace provides a beautiful answer to a difficult question, turning a messy chemical separation problem into a clean, predictable physical one. Achieving this level of control requires equipment built for precision and reliability. The integrity of the vacuum seal, the accuracy of the temperature profile, and the robustness of the system are not just features; they are the foundation of the entire process.
This is where purpose-built systems, like the customizable vacuum furnaces from KINTEK, become critical. Engineered to deliver the precise control and deep vacuum required, they provide the reliable foundation for efficient and high-purity metal recovery.
| Key Process Parameter | Typical Value / Condition | Purpose |
|---|---|---|
| Vacuum Pressure | Below 10 Pa | Lowers magnesium's boiling point for efficient vaporization. |
| Operating Temperature | ~700°C | Provides thermal energy for phase change without vaporizing impurities. |
| Primary Impurities | Aluminum, Iron, Silicon | Left behind as solids/liquids due to lower vapor pressure. |
| Recovery Mechanism | Distillation & Condensation | Magnesium vapor condenses on a cooler surface into pure solid form. |
To see how a precision-engineered furnace can transform your recovery operations, Contact Our Experts.
Visual Guide
Related Products
- Magnesium Extraction and Purification Condensing Tube Furnace
- Vacuum Heat Treat Sintering Furnace with Pressure for Vacuum Sintering
- Small Vacuum Heat Treat and Tungsten Wire Sintering Furnace
- 2200 ℃ Tungsten Vacuum Heat Treat and Sintering Furnace
- Vacuum Heat Treat Sintering Furnace Molybdenum Wire Vacuum Sintering Furnace
Related Articles
- The Controlled Universe: Why Tube Furnaces Prioritize Precision Over Scale
- Beyond Heat: Mastering the Controlled Universe Inside a Tube Furnace
- Your Furnace Isn't Just a Heater: Why 'Good Enough' Equipment Is Sabotaging Your Advanced Materials Research
- Mastering the Micro-Environment: The Unseen Power of the Tube Furnace
- The Art of Isolation: Mastering Material Properties with Tube Furnaces




















