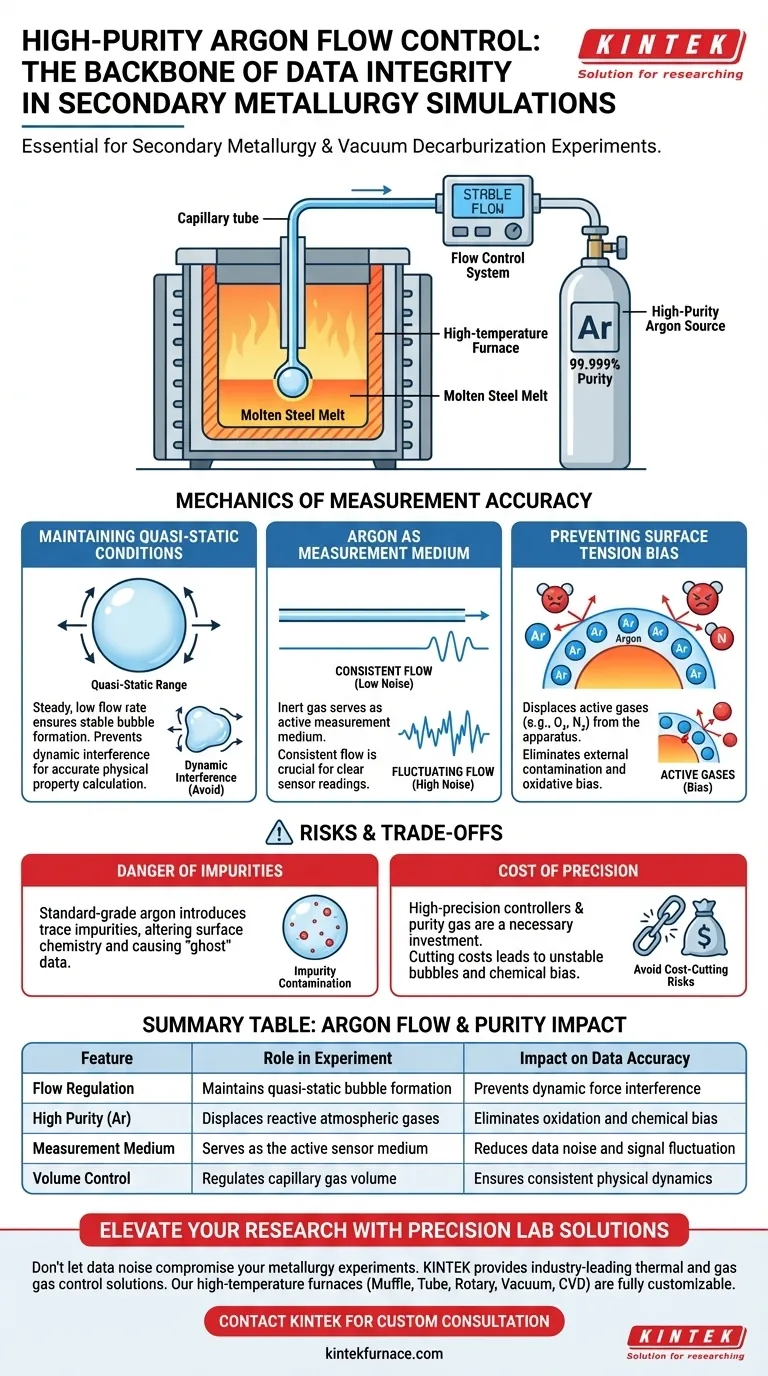
A high-purity argon flow control system is the backbone of data integrity in secondary metallurgy and vacuum decarburization simulations. Its primary function is to regulate the precise volume of gas flowing into the capillary, ensuring that the physical dynamics of the experiment remain constant. Without this strict control, both the mechanical formation of bubbles and the chemical purity of the steel melt would be compromised, rendering surface tension measurements invalid.
Precise argon control is the only way to maintain the quasi-static bubble formation necessary for accurate measurement while simultaneously displacing active gases that would otherwise bias data through oxidation.

The Mechanics of Measurement Accuracy
Maintaining Quasi-Static Conditions
In simulation experiments, the rate at which gas bubbles form is a critical variable. The flow control system ensures the volume flow into the capillary is low and steady enough to keep bubble formation in the quasi-static range.
If the flow exceeds this range, dynamic forces interfere with the bubble shape. This interference prevents accurate calculation of the melt's physical properties.
Argon as the Measurement Medium
The inert gas is not just a protective blanket; it serves as the active measurement medium.
The consistency of the argon flow directly impacts the sensor readings. Fluctuations in flow create noise in the data, obscuring the subtle surface tension changes you are trying to measure.
Preventing Surface Tension Bias
Surface tension measurements are highly sensitive to surface chemistry. The flow control system ensures a continuous supply of high-purity argon to displace active gases within the apparatus.
By removing reactive elements like oxygen from the environment, the system prevents external contamination. This ensures the measured surface tension reflects the true properties of the steel melt, not a reaction with the atmosphere.
Understanding the Risks and Trade-offs
The Danger of Impurities
While flow control is essential, the purity of the gas is equally critical. Even with perfect flow control, using standard-grade argon can introduce trace impurities.
These impurities can alter the surface chemistry of the melt. This results in "ghost" data where the measurements are precise (consistent) but inaccurate (biased by oxidation).
The Cost of Precision
High-precision flow controllers and high-purity gas sources represent a significant investment.
However, the trade-off is unavoidable. attempting to save costs by using rougher flow metering or lower-grade gas will likely result in bubble instability and chemical bias, requiring the experiment to be repeated.
Ensuring Experimental Success
To guarantee valid results in your secondary metallurgy simulations, align your equipment choices with your specific data requirements:
- If your primary focus is precise surface tension data: Prioritize a flow controller capable of maintaining strict stability within the quasi-static bubble formation range.
- If your primary focus is melt chemistry and purity: Ensure your system uses verified high-purity argon to eliminate any risk of oxidative bias.
The integrity of your simulation data is defined by the stability and purity of the atmosphere you create around the melt.
Summary Table:
| Feature | Role in Experiment | Impact on Data Accuracy |
|---|---|---|
| Flow Regulation | Maintains quasi-static bubble formation | Prevents dynamic force interference |
| High Purity (Ar) | Displaces reactive atmospheric gases | Eliminates oxidation and chemical bias |
| Measurement Medium | Serves as the active sensor medium | Reduces data noise and signal fluctuation |
| Volume Control | Regulates capillary gas volume | Ensures consistent physical dynamics |
Elevate Your Research with Precision Lab Solutions
Don't let data noise or oxidative bias compromise your metallurgy experiments. KINTEK provides industry-leading thermal and gas control solutions backed by expert R&D and manufacturing.
Whether you require Muffle, Tube, Rotary, Vacuum, or CVD systems, our high-temperature furnaces are fully customizable to your unique research needs. We ensure your simulation environment maintains the stability and purity required for high-integrity results.
Ready to optimize your experimental setup? Contact KINTEK today for a custom consultation!
Visual Guide
References
- Lukas Neubert, Olena Volkova. Effect of Oxygen on Thermophysical Properties of Molten High-Silicon Electrical Steels and Its Impact on Bubble Formation Behavior. DOI: 10.1007/s11663-025-03594-9
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- 1200℃ Controlled Inert Nitrogen Atmosphere Furnace
- Magnesium Extraction and Purification Condensing Tube Furnace
- Electric Rotary Kiln Pyrolysis Furnace Plant Machine Small Rotary Kiln Calciner
- CF KF Flange Vacuum Electrode Feedthrough Lead Sealing Assembly for Vacuum Systems
- Laboratory Muffle Oven Furnace with Bottom Lifting
People Also Ask
- What is the use of nitrogen in furnace? Prevent Oxidation for Superior Heat Treatment
- What are the benefits of inert atmosphere heat treating? Prevent Oxidation and Preserve Material Integrity
- How does an inert atmosphere prevent oxidation? Shield Materials from Oxygen Damage
- How does nitrogen atmosphere heat treatment improve surface strengthening? Enhance Durability and Performance
- What is the main purpose of heat treatment? Transform Metal Properties for Superior Performance



















