The Illusion of Force
When separating metals, our intuition gravitates toward what we can see and feel: intense heat. We assume that applying more energy—cranking up the temperature—is the primary driver of success.
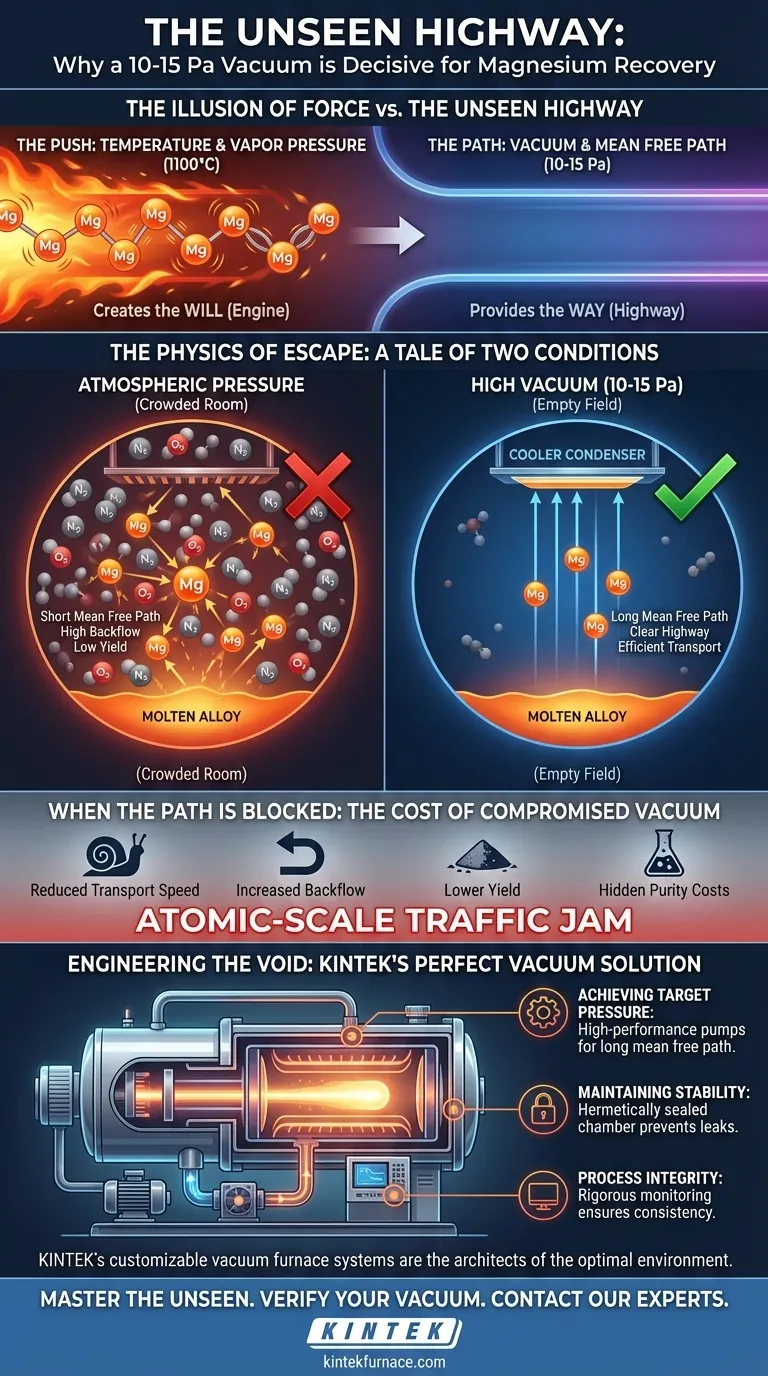
This is a powerful psychological bias. We focus on the brute force of heat making magnesium want to evaporate from an aluminum-magnesium alloy. But we often overlook the more subtle, and ultimately more critical, factor: the environment that allows it to escape.
Temperature provides the will. A high vacuum provides the way.
The Physics of Escape: A Tale of Two Conditions
To efficiently separate magnesium, two conditions must be met. One is about impetus, the other about opportunity. Confusing them is the most common source of failure.
The Push: Temperature and Vapor Pressure
At 1100 °C (1373 K), magnesium atoms within the molten alloy are vibrating with tremendous energy. This gives them a high saturated vapor pressure—a strong thermodynamic drive to break their metallic bonds and transition into a gaseous state.
This is the easy part to understand. It's the engine of the process.
The Path: Vacuum and the Mean Free Path
The true bottleneck is not creating the vapor, but transporting it. This is where a high-vacuum environment of 10-15 Pa becomes the hero of the story.
By evacuating the furnace, we remove the air and other residual gas molecules. This dramatically extends the mean free path—the average distance a magnesium vapor molecule can travel before colliding with another particle.
Think of it as the difference between running across a crowded stadium concourse versus an empty field.
- Atmospheric Pressure: A chaotic, crowded room. Magnesium vapor collides constantly, losing energy and scattering randomly. Many atoms are knocked back into the molten alloy.
- High Vacuum (10-15 Pa): An open, empty field. Magnesium vapor travels in a nearly straight line, directly from the alloy's surface to the cooler condenser.
The vacuum doesn't push the magnesium. It simply clears the highway for it.
When the Path is Blocked: The High Cost of a Compromised Vacuum
Failing to achieve or maintain the target vacuum transforms an elegant separation process into a slow, inefficient struggle. The consequences are immediate and severe.
A Traffic Jam at the Atomic Scale
When the pressure is too high, the mean free path shrinks. The result is an atomic-scale traffic jam.
Volatilized magnesium atoms collide with nitrogen, oxygen, and other molecules. This chaos throttles the entire system:
- Reduced Transport Speed: The vapor's journey to the condenser slows to a crawl.
- Increased Backflow: A significant portion of the vapor is deflected back toward the molten alloy, re-dissolving and effectively lost.
- Lower Yield: Fewer magnesium atoms ever reach the collection point, directly crippling the recovery rate.
The Hidden Costs in Purity
The issue isn't just about lost material. A poor vacuum also introduces contaminants. The residual gases can react with the highly energetic magnesium vapor or interfere with the condensation process, leading to a final product of lower purity.
Engineering the Void: The Practicalities of a Perfect Vacuum
Creating an environment that is almost a complete absence of matter is a profound engineering challenge. It demands more than just a good pump; it requires a perfectly integrated system.
| Challenge | Engineering Requirement | Impact on Process |
|---|---|---|
| Achieving Target Pressure | High-performance vacuum pumps (e.g., rotary, diffusion, molecular) | Creates the necessary long mean free path. |
| Maintaining Stability | A robust, hermetically sealed furnace chamber | Prevents leaks that would cause pressure fluctuations and kill efficiency. |
| Process Integrity | Rigorous monitoring and high-quality components | Ensures consistency and repeatability, run after run. |
This is where the design and manufacturing precision of a furnace system becomes the bedrock of process reliability. Achieving a stable 10-15 Pa environment isn't accidental; it's the result of meticulous engineering found in specialized equipment, such as KINTEK's customizable vacuum furnace systems. These systems are designed not just to heat, but to create and protect the pristine vacuum conditions essential for success.
Mastering the Unseen
Ultimately, the success of separating aluminum-magnesium alloys hinges on controlling this invisible highway. The most common cause of low yield isn't a lack of heat, but a compromised path.
Before questioning your temperatures or materials, always verify the vacuum.
- Is your system reaching the target 10-15 Pa?
- Is it holding that pressure stably throughout the process?
- Has the system been checked for even the smallest leaks?
The right furnace is not merely a container for the reaction; it is the architect of the optimal environment. To ensure your process benefits from a stable, high-performance vacuum tailored to your specific material needs, Contact Our Experts.
Visual Guide
Related Products
- Magnesium Extraction and Purification Condensing Tube Furnace
- Molybdenum Vacuum Heat Treat Furnace
- Vacuum Heat Treat Sintering Furnace Molybdenum Wire Vacuum Sintering Furnace
- 2200 ℃ Tungsten Vacuum Heat Treat and Sintering Furnace
- 2200 ℃ Graphite Vacuum Heat Treat Furnace
Related Articles
- From Dust to Density: Mastering Material Transformation in a Tube Furnace
- Mastering the Void: How Tube Furnaces Forge the Atoms of Innovation
- Your Furnace Isn't Just a Heater: Why 'Good Enough' Equipment Is Sabotaging Your Advanced Materials Research
- Mastering the Void: How Tube Furnaces Engineer Predictable Outcomes
- Beyond Heat: Mastering the Controlled Universe Inside a Tube Furnace




















