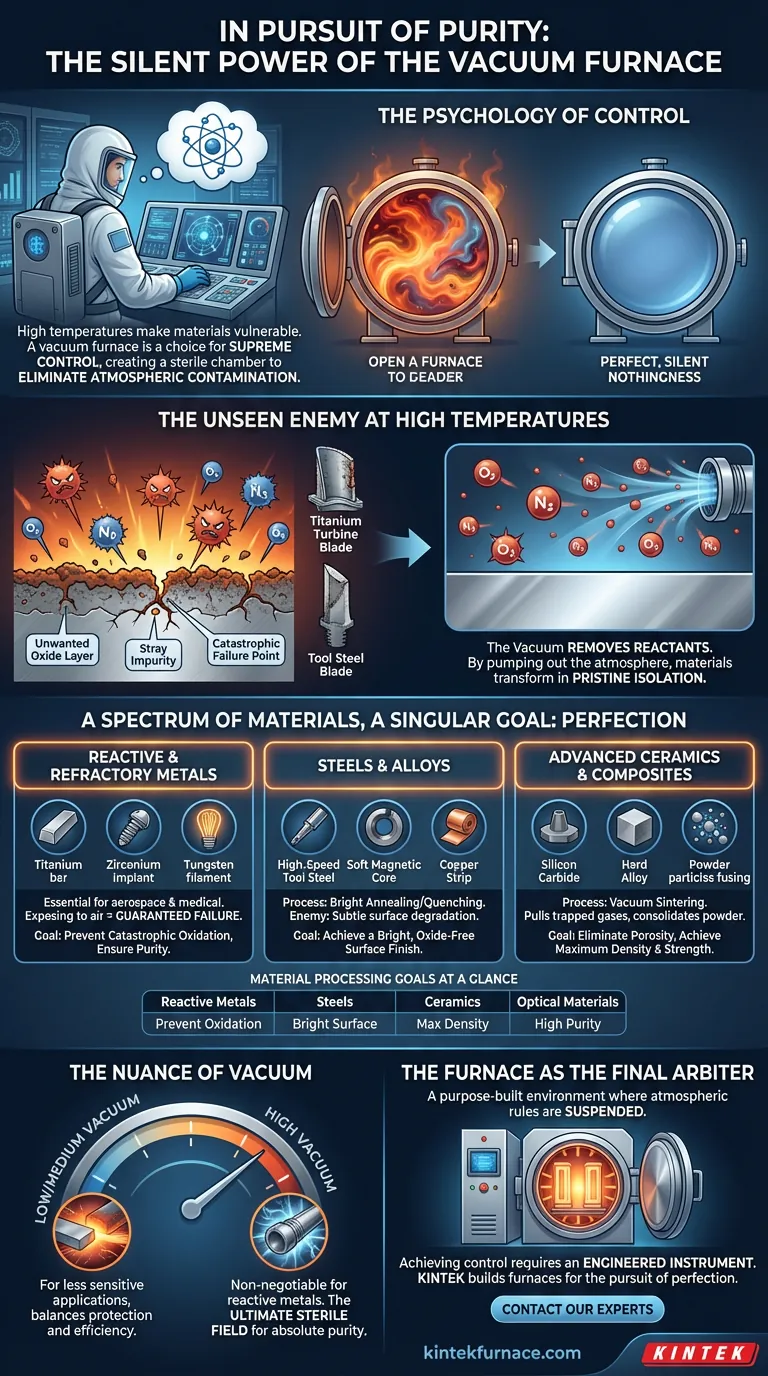
The Psychology of Control
At high temperatures, materials become vulnerable. Their atomic structures loosen, making them receptive to transformation but also susceptible to attack. For a materials engineer, this is a moment of both supreme opportunity and profound risk.
The decision to use a vacuum furnace stems from a fundamental human and scientific desire: control. It is an admission that the air we breathe, the invisible and life-giving atmosphere, becomes a contaminant—an enemy—in the crucible of creation.
The furnace isn't just a tool for heating; it's a sterile chamber designed to eliminate the variable of chemistry itself. It's where we impose our will on matter by first creating a perfect, silent nothingness.
The Unseen Enemy at High Temperatures
Most failures in materials science don't come from grand, visible flaws. They begin with microscopic imperfections—an unwanted oxide layer, a stray impurity, a tiny void. At high temperatures, the oxygen and nitrogen in the atmosphere are aggressive reactants, eager to bond with and corrupt a material's surface.
This is more than just rust. For a titanium turbine blade, an oxide inclusion can become a stress point, leading to catastrophic failure. For a high-speed steel tool, a surface scale dulls its cutting edge before it ever makes a cut.
The vacuum's function is simple but absolute: it removes the reactants. By pumping out the atmosphere, it removes the threat, allowing the material to undergo its transformation in a state of pristine isolation.
A Spectrum of Materials, A Singular Goal: Perfection
While the materials vary, the goal is always to protect or perfect a specific property that would otherwise be compromised by atmospheric exposure.
The Untouchables: Reactive and Refractory Metals
This is the classic domain of the vacuum furnace. Metals prized for their extreme performance—like titanium, tungsten, and zirconium—are also exceptionally reactive at high temperatures. Exposing them to air while heating is not an option; it's a guarantee of failure.
- Titanium (Ti) & Zirconium (Zr): Essential for aerospace and medical implants, their integrity cannot be compromised.
- Tungsten (W) & Molybdenum (Mo): Used in high-temperature applications, any oxidation degrades their performance.
Processing these metals in a vacuum isn't a best practice; it is the only practice.
The Pursuit of a Flawless Surface: Steels and Alloys
For many alloys, the enemy is not catastrophic failure but a subtle degradation of the surface. Processes like "bright annealing" and "bright quenching" are named for their objective: a clean, shiny, oxide-free finish.
- High-Speed Tool Steel & Die Steel: Require a perfect surface to maintain hardness and sharpness.
- Soft Magnetic Alloys: Need extreme purity to achieve their delicate magnetic properties.
- Copper Strips & Aluminum Foil: A clean surface is critical for subsequent processing and performance.
Here, the vacuum acts as a guardian, preventing the formation of scale that would otherwise need to be cleaned off through costly and often damaging secondary processes.
Forging Density from Dust: Advanced Ceramics and Composites
Vacuum sintering is a process of creation. It takes powdered materials and fuses them into a solid, high-density part. The vacuum plays a dual role here.
First, it prevents reactions. Second, and just as critically, it pulls trapped gases out from between the powder particles. This allows the material to consolidate fully, eliminating voids and creating a final product—like Silicon Carbide (SiC) or hard alloys—with superior strength and density.
Not All Silence is Equal: The Nuance of Vacuum
The pursuit of control comes in degrees. The level of vacuum required is a strategic decision, balancing the material's sensitivity against the cost and complexity of the process.
-
High Vacuum: For the most reactive metals like titanium, a high vacuum is non-negotiable. It's the ultimate sterile field, removing the maximum number of atmospheric molecules to ensure absolute purity.
-
Low/Medium Vacuum: For less sensitive applications, like annealing certain steels, a lower vacuum may be sufficient. It provides enough protection to prevent significant oxidation while being more efficient to achieve.
This choice reflects a deep understanding of the material and the process. It's about applying the precise level of control needed—no more, no less.
Material Processing Goals at a Glance
| Material Category | Examples | Key Process Objective |
|---|---|---|
| Reactive & Refractory Metals | Titanium, Zirconium, Tungsten | Prevent catastrophic oxidation, ensure purity |
| Steels & Specialty Alloys | High-speed tool steel, Soft magnetic alloys | Achieve a bright, oxide-free surface finish |
| Advanced Ceramics & Composites | Silicon carbide (SiC), Hard alloys (cermets) | Eliminate porosity, achieve maximum density/strength |
| Optical & Infrared Materials | Zinc sulfide (ZnS), Zinc selenide (ZnSe) | Synthesize high-purity compounds for clarity |
The Furnace as the Final Arbiter
Ultimately, a vacuum furnace is more than an oven. It is a purpose-built environment, a small pocket of the universe where the rules of atmospheric chemistry are temporarily suspended. Choosing to use one is a statement about the standards you hold for your material's final form.
Achieving this level of control requires not just any furnace, but one engineered for the specific demands of your material and process. At KINTEK, we build the instruments for this pursuit of perfection. Our range of customizable Muffle, Tube, Vacuum, and CVD furnaces are designed to provide the precise, stable, and pure high-temperature environments your research demands. We understand that every experiment is a quest for a specific outcome, and our systems are built to ensure you can achieve it.
When the integrity of your material cannot be compromised, creating the perfect vacuum is the first and most critical step. Contact Our Experts
Visual Guide
Related Products
- Vacuum Hot Press Furnace Machine Heated Vacuum Press Tube Furnace
- Vacuum Hot Press Furnace Machine Heated Vacuum Press
- 600T Vacuum Induction Hot Press Vacuum Heat Treat and Sintering Furnace
- 2200 ℃ Tungsten Vacuum Heat Treat and Sintering Furnace
- Vacuum Heat Treat Sintering Furnace with Pressure for Vacuum Sintering
Related Articles
- Beyond the Parts List: The Real Reason Your Vacuum Hot Press Fails (And How to Fix It)
- More Than a Void: The Inherent Energy Efficiency of Vacuum Furnace Design
- The Pressure Spectrum: Why a Vacuum Furnace's Capability Isn't a Single Number
- The Unseen Forces in a Vacuum Furnace: A Discipline of Control
- Is Your Material Data Real? Why Temperature Gradients Are Sabotaging Your Cu2Se Resistivity Measurements




















