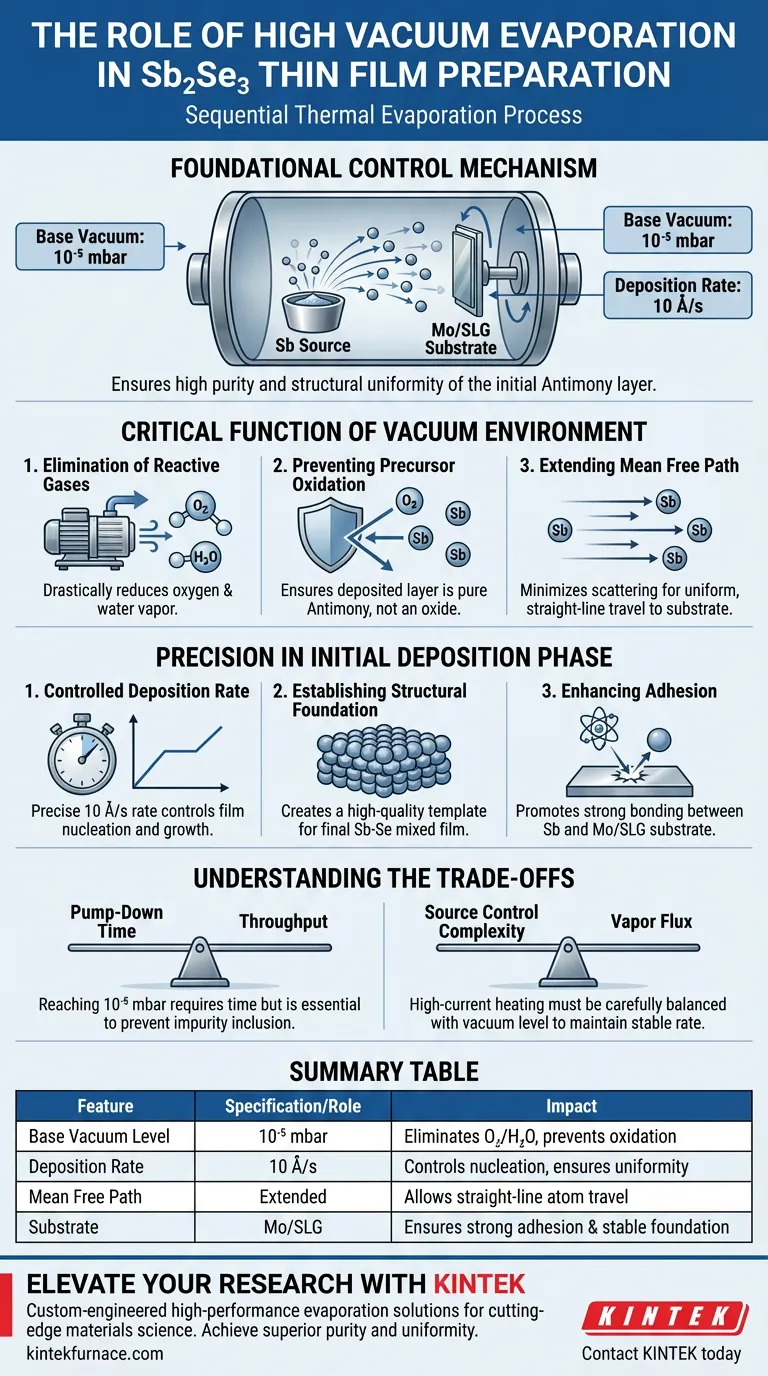
The high vacuum evaporation system serves as the foundational control mechanism for the sequential preparation of Sb2Se3 thin films. Its primary role is to establish a base vacuum of 10⁻⁵ mbar, enabling the Antimony (Sb) element source to be deposited onto Molybdenum/Soda-Lime Glass (Mo/SLG) substrates at a stable rate of 10 Å/s without environmental interference.
The system ensures that the initial antimony layer—the precursor for the final compound—is deposited with high purity and structural uniformity. This step is critical because any oxidation or instability at this stage will compromise the composition and performance of the final Sb-Se absorber layer.
The Critical Function of the Vacuum Environment
Elimination of Reactive Gases
The most immediate function of the high vacuum system is the removal of atmospheric contaminants. By reaching a base pressure of 10⁻⁵ mbar, the system drastically reduces the presence of oxygen and water vapor.
Preventing Precursor Oxidation
During the heating of the Antimony (Sb) source, the material is highly susceptible to reaction. The vacuum environment prevents the oxidation of the metal vapor during its transit from the source to the substrate. This ensures the deposited layer remains pure Antimony, rather than an oxide, which is essential for the subsequent reaction with Selenium.
Extending the Mean Free Path
While the primary focus is purity, the vacuum also governs the physics of travel. A high vacuum minimizes collisions between evaporated Sb atoms and residual gas molecules. This allows the atoms to travel in a straight-line path to the substrate, preventing scattering that could lead to uneven coverage.
Precision in the Initial Deposition Phase
Controlled Deposition Rate
The system allows for precise thermal regulation, enabling a specific deposition rate of 10 Å/s. Maintaining this stable rate is vital for controlling the nucleation and growth of the film.
Establishing the Structural Foundation
The initial Sb layer acts as the physical template for the final thin film. By ensuring this layer is deposited uniformly on the Mo/SLG substrate, the system creates a high-quality physical foundation. This uniformity allows for controllable composition when the layer is later processed into the final Sb-Se mixed thin film.
Enhancing Adhesion
The lack of gaseous interference ensures the Sb atoms arrive at the substrate with sufficient kinetic energy. This promotes strong adhesion between the Antimony layer and the Molybdenum-coated glass, preventing delamination during subsequent thermal processing steps.
Understanding the Trade-offs
Pump-Down Time vs. Throughput
Achieving a vacuum level of 10⁻⁵ mbar or better requires significant pumping time, which can limit manufacturing throughput. Rushing this stage (e.g., stopping at 10⁻⁴ mbar) significantly increases the risk of oxidation and impurity inclusion.
Source Control Complexity
While the vacuum enables purity, it complicates the control of vapor flux. High-current heating in a vacuum can lead to rapid evaporation spikes if not carefully managed. You must balance the heating current against the vacuum level to maintain the target 10 Å/s rate without overwhelming the substrate.
Making the Right Choice for Your Goal
To optimize your Sb2Se3 thin film preparation, consider the following priorities:
- If your primary focus is Film Purity and Efficiency: Ensure your system consistently reaches a base pressure of 10⁻⁵ mbar before heating to prevent oxide barriers that impede charge transport.
- If your primary focus is Structural Uniformity: Prioritize the stability of the deposition rate (10 Å/s) and substrate rotation to ensure the precursor layer has no thickness gradients.
The quality of your final Sb2Se3 device is determined by the purity of the initial Antimony deposition, making the high vacuum environment a non-negotiable requirement for high-performance absorbers.
Summary Table:
| Feature | Specification/Role | Impact on Sb2Se3 Thin Film |
|---|---|---|
| Base Vacuum Level | 10⁻⁵ mbar | Eliminates oxygen/water vapor; prevents precursor oxidation. |
| Deposition Rate | 10 Å/s | Controls nucleation and ensures structural uniformity. |
| Mean Free Path | Extended (High Vacuum) | Allows straight-line atom travel for even substrate coverage. |
| Substrate Compatibility | Mo/SLG (Molybdenum/Glass) | Ensures strong adhesion and a stable structural foundation. |
| Atmospheric Control | Removal of Reactive Gases | Maintains high chemical purity of the Antimony (Sb) layer. |
Elevate Your Research with Precision Vacuum Systems
At KINTEK, we understand that the quality of your Sb2Se3 absorbers depends on the integrity of your vacuum environment. Our high-performance evaporation solutions are engineered to eliminate contamination and provide the stable deposition rates necessary for high-efficiency thin film solar cells.
Why choose KINTEK?
- Expert R&D & Manufacturing: Custom-engineered systems designed for cutting-edge materials science.
- Versatile Thermal Solutions: From Muffle and Tube furnaces to specialized Vacuum, CVD, and Rotary systems.
- Tailored for You: All systems are fully customizable to meet your specific laboratory or industrial needs.
Ready to achieve superior film purity and structural uniformity? Contact KINTEK today to discuss how our customizable high-temperature systems can optimize your fabrication process.
Visual Guide
References
- Maykel Jiménez-Guerra, Edgardo Saucedo. KCN Chemical Etching of van der Waals Sb<sub>2</sub>Se<sub>3</sub> Thin Films Synthesized at Low Temperature Leads to Inverted Surface Polarity and Improved Solar Cell Efficiency. DOI: 10.1021/acsaem.3c01584
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Custom Made Versatile CVD Tube Furnace Chemical Vapor Deposition CVD Equipment Machine
- 2200 ℃ Tungsten Vacuum Heat Treat and Sintering Furnace
- Stainless Steel Quick Release Vacuum Chain Three Section Clamp
- Vacuum Heat Treat Sintering Furnace Molybdenum Wire Vacuum Sintering Furnace
- High Pressure Laboratory Vacuum Tube Furnace Quartz Tubular Furnace
People Also Ask
- What functions does glucose perform in lithium-ion sieve synthesis? Enhance Carbothermal Reduction for LiMnO2 Purity
- What is the working principle of a CVD tube furnace? Achieve Precise Thin Film Deposition for Your Lab
- Why is the tube design important in CVD furnaces? Ensure Uniform Deposition for High-Quality Films
- What advantages does the Laser Chemical Vapor Deposition (LCVD) process offer? High Purity & Precision SiC Fibers
- What is the function of injecting water in wood thermal modification? Unlock Superior Stability and Hydrophobicity



















