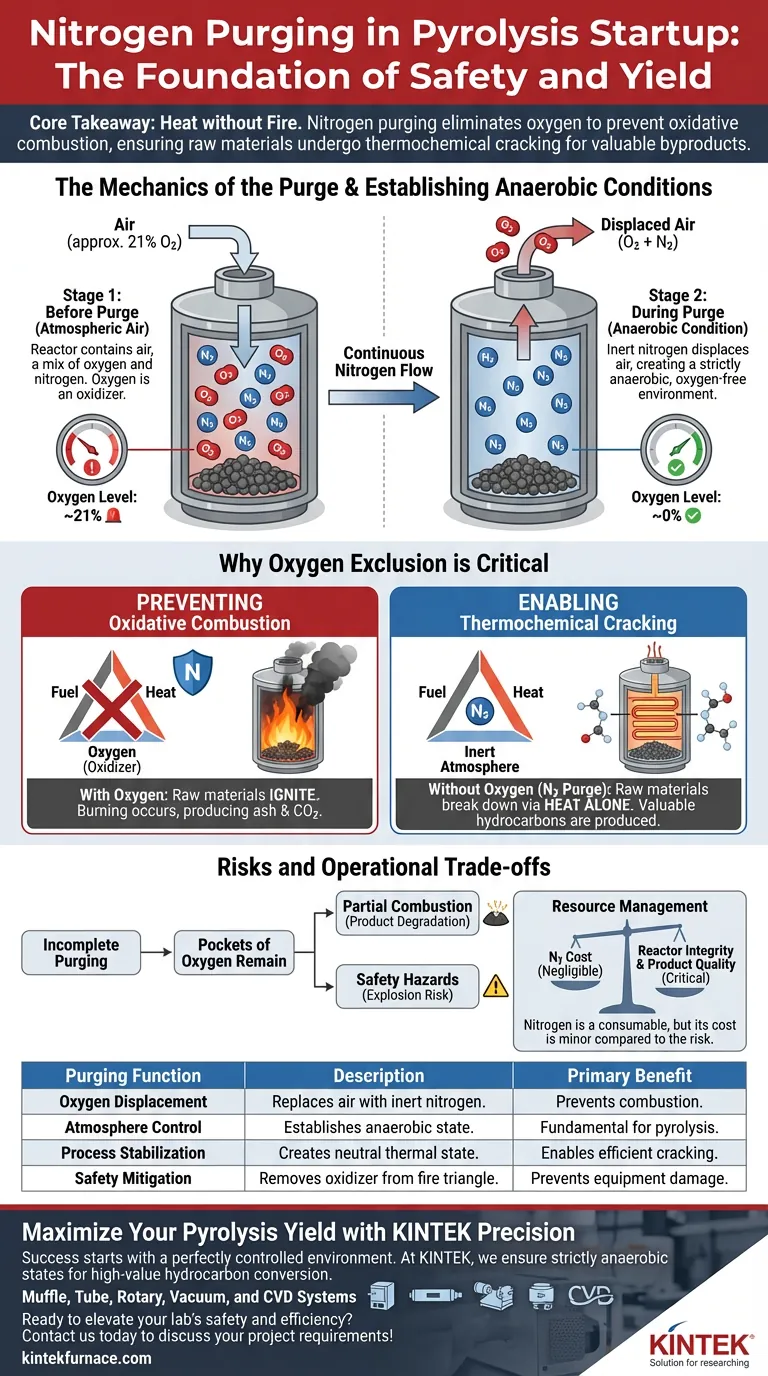
Nitrogen purging is the definitive safety and process initiation step required before bringing a pyrolysis reactor up to temperature. Its primary function is to systematically displace atmospheric air within the reaction chamber to create a completely oxygen-free environment.
By introducing a continuous flow of nitrogen, the system ensures that the raw materials are subjected to heat in a strictly anaerobic state, which is the fundamental requirement for successful pyrolysis.
Core Takeaway Pyrolysis requires heat without fire. Nitrogen purging eliminates oxygen to prevent the raw materials from burning (oxidative combustion), ensuring they instead undergo thermochemical cracking to produce the desired chemical byproducts.

The Mechanics of the Purge
Displacing the Reactor Atmosphere
Before startup, the reactor chamber contains atmospheric air, which is roughly 21% oxygen.
To remove this, nitrogen is introduced into the system continuously.
This flow physically pushes the existing air out of the chamber, replacing it volume-for-volume with inert nitrogen gas.
Establishing Anaerobic Conditions
The goal is to achieve a state where the oxygen content is effectively zero.
This state is referred to as an anaerobic condition.
Without achieving this specific environment, the chemical process defined as pyrolysis cannot technically occur.
Why Oxygen Exclusion is Critical
Preventing Oxidative Combustion
If oxygen remains in the chamber when the reactor heats up, the raw materials will ignite.
This reaction is known as oxidative combustion, essentially burning the material rather than breaking it down.
Nitrogen acts as a safety blanket, removing the oxidizer (oxygen) from the fire triangle so that combustion is physically impossible, regardless of the temperature.
Enabling Thermochemical Cracking
Pyrolysis depends on thermochemical cracking—breaking long-chain molecules into smaller ones using heat alone.
This delicate chemical reaction requires a neutral atmosphere.
Nitrogen ensures the environment is stable enough for this cracking to occur efficiently, preserving the quality of the end products.
Risks and Operational Trade-offs
The Consequence of Incomplete Purging
If the purging phase is rushed or incomplete, pockets of oxygen may remain.
This can lead to partial combustion, which degrades the final product (turning valuable hydrocarbons into ash and CO2).
In severe cases, introducing high heat to a mix of flammable gases and residual oxygen can pose significant safety hazards.
Resource Management
Nitrogen is a consumable cost during the startup phase.
Operators must balance the time and volume of nitrogen used against the absolute necessity of a safe atmosphere.
However, the cost of nitrogen is always negligible compared to the risk of compromising the reactor integrity or the quality of the batch.
Ensuring Process Success
To maximize the efficiency and safety of your pyrolysis startup, evaluate your purging strategy based on your specific operational goals.
- If your primary focus is Safety: Verify that oxygen levels are undetectable before activating heating elements to eliminate combustion risks.
- If your primary focus is Product Quality: Ensure the environment is strictly anaerobic to guarantee thermochemical cracking maximizes yield rather than creating ash.
A disciplined nitrogen purge is the invisible foundation of a high-yield, safe pyrolysis operation.
Summary Table:
| Purging Function | Description | Primary Benefit |
|---|---|---|
| Oxygen Displacement | Replaces atmospheric air with inert nitrogen. | Prevents oxidative combustion/burning. |
| Atmosphere Control | Establishes a strictly anaerobic environment. | Fundamental requirement for pyrolysis. |
| Process Stabilization | Creates a neutral state for thermal reactions. | Enables efficient thermochemical cracking. |
| Safety Mitigation | Removes the oxidizer from the fire triangle. | Prevents explosions and equipment damage. |
Maximize Your Pyrolysis Yield with KINTEK Precision
Success in pyrolysis starts with a perfectly controlled environment. At KINTEK, we understand that maintaining a strictly anaerobic state is critical for converting raw materials into high-value hydrocarbons without the risk of combustion.
Backed by expert R&D and world-class manufacturing, KINTEK offers a comprehensive range of laboratory high-temperature furnaces, including Muffle, Tube, Rotary, Vacuum, and CVD systems. Whether you require standard equipment or a solution fully customizable for your unique thermochemical cracking needs, our technical team is ready to support your research and production goals.
Ready to elevate your lab's safety and efficiency?
Contact us today to discuss your project requirements!
Visual Guide
References
- Ana B. Cuevas, M.P. Dorado. An Overview of Pyrolysis as Waste Treatment to Produce Eco-Energy. DOI: 10.3390/en17122852
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- 1700℃ Controlled Inert Nitrogen Atmosphere Furnace
- 1400℃ Controlled Inert Nitrogen Atmosphere Furnace
- 1200℃ Controlled Inert Nitrogen Atmosphere Furnace
- Mesh Belt Controlled Atmosphere Furnace Inert Nitrogen Atmosphere Furnace
- Controlled Inert Nitrogen Hydrogen Atmosphere Furnace
People Also Ask
- Why is electromagnetic stirring necessary for NbC-Cr7C3@graphene in W18Cr4V steel? Boost Alloy Performance
- What are the key features of an atmosphere box furnace? Unlock Precise Heat Processing in Controlled Environments
- What is the function of vacuum & heating in aluminum degassing? Enhancing Composite Integrity & Density
- What is inert gas technology used for in high-temperature atmosphere vacuum furnaces? Protect Materials and Speed Up Cooling
- What is the function of injecting water in wood thermal modification? Unlock Superior Stability and Hydrophobicity



















