The Paradox of a Vacuum Furnace
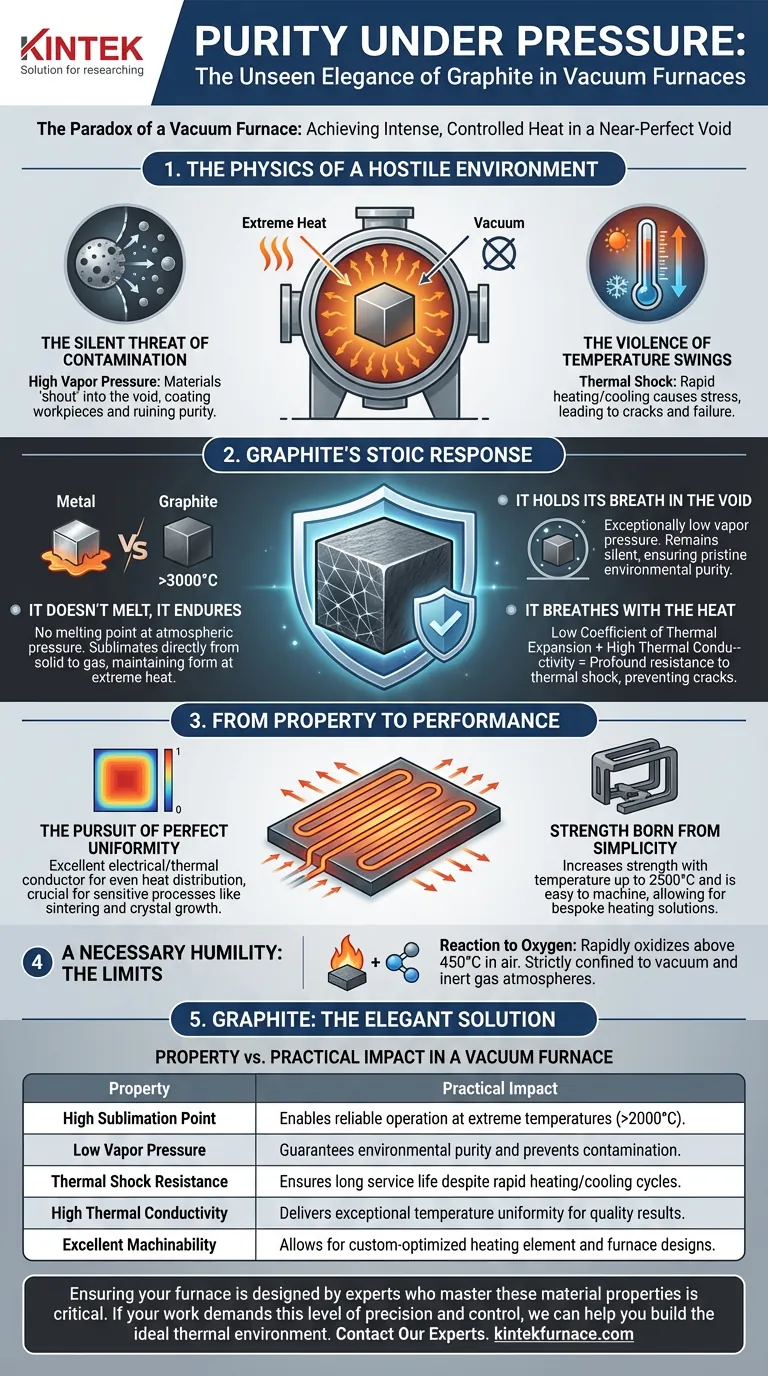
A high-temperature vacuum furnace is a place of profound contradiction. It's an environment engineered to be empty, a near-perfect void. Yet, within that void, we must generate immense energy—heat so intense it can transform the very structure of materials.
This creates a brutal engineering challenge. Most materials, when faced with extreme heat, begin to fight back. They melt, they warp, and, most critically in a vacuum, they "outgas"—shedding molecules and contaminating the pristine environment you worked so hard to create.
The goal is not just heat, but controlled heat. It's about achieving purity under pressure. This is where the quiet, unassuming elegance of graphite comes into play.
The Physics of a Hostile Environment
To appreciate graphite's role, you must first appreciate the hostility of the environment it tames. A high-temperature vacuum is unforgiving.
The Silent Threat of Contamination
In a vacuum, a material's vapor pressure becomes its voice. Materials with high vapor pressure shout into the void, releasing particles that coat your workpiece and ruin the process. This isn't just a mess; it's a fundamental failure of control. You lose the chemical purity essential for advanced materials science.
The Violence of Temperature Swings
Industrial processes demand speed. Furnaces must heat up and cool down rapidly. This thermal shock induces incredible stress on materials, like repeatedly bending a paperclip. Most will eventually crack and fail, leading to downtime and costly repairs.
This is the central problem: How do you find a material that can get incredibly hot without contaminating the void, and endure violent temperature changes without breaking?
Graphite’s Stoic Response
Graphite doesn't just survive in this environment; it thrives. It solves these problems not through brute force, but through its fundamental physical nature.
It Doesn't Melt, It Endures
Most metals have a clear breaking point—a melting temperature. Graphite doesn't. At atmospheric pressure, it has no melting point at all. Instead, at temperatures above 3000°C—far beyond the operational range of most industrial metals—it sublimates, turning directly from a solid to a gas.
This property gives it an almost surreal level of stability. It holds its form and function at temperatures that would turn tungsten or molybdenum into liquid.
It Holds Its Breath in the Void
Graphite possesses an exceptionally low vapor pressure. In the face of intense heat, it remains silent. It does not shed particles or contaminate the vacuum. This ensures the process environment remains as pure as intended, allowing for predictable, repeatable results.
This isn't just a feature; it's the foundation of trust in your process.
It Breathes with the Heat
Confronted with rapid temperature changes, graphite remains unfazed. This is due to a powerful combination of two properties:
- Low Coefficient of Thermal Expansion: It barely expands or contracts when heated or cooled.
- High Thermal Conductivity: It dissipates thermal stress quickly and evenly across its structure.
Together, these traits give graphite a profound resistance to thermal shock. It accepts the stress of rapid cycles without cracking, ensuring a long and reliable service life.
From Property to Performance
These physical characteristics translate directly into superior operational performance, which is what engineers and scientists truly care about.
The Pursuit of Perfect Uniformity
Graphite is an excellent electrical and thermal conductor. When current passes through it, heat is generated efficiently and, more importantly, spreads evenly.
This results in outstanding temperature uniformity across the furnace's hot zone. For sensitive processes like sintering, brazing, or crystal growth, this uniformity isn't a luxury; it's the difference between success and failure.
Strength Born from Simplicity
Despite being lightweight, graphite is mechanically strong, and its strength actually increases with temperature up to 2500°C. Furthermore, it is remarkably easy to machine.
This allows for the creation of intricate and complex heating elements, custom-designed to optimize heat distribution for any furnace geometry. This is where standardized solutions fall short and bespoke engineering, like that offered by KINTEK, provides a decisive advantage. Their ability to customize vacuum and CVD furnace systems leverages this very property to meet unique process demands.
| Property | Practical Impact in a Vacuum Furnace |
|---|---|
| High Sublimation Point | Enables reliable operation at extreme temperatures (>2000°C). |
| Low Vapor Pressure | Guarantees environmental purity and prevents contamination. |
| Thermal Shock Resistance | Ensures long service life despite rapid heating/cooling cycles. |
| High Thermal Conductivity | Delivers exceptional temperature uniformity for quality results. |
| Excellent Machinability | Allows for custom-optimized heating element and furnace designs. |
A Necessary Humility: The Limits
No material is a silver bullet. Graphite's strength is tied to its environment. Its one major limitation is its reaction to oxygen.
At temperatures above 450°C, graphite will rapidly oxidize (burn) in the presence of air. This is why its dominance is strictly confined to the worlds of vacuum and inert gas atmospheres.
For the small subset of applications that are hyper-sensitive to even trace amounts of carbon, more expensive refractory metals may be a necessary alternative. But for the vast majority of high-temperature vacuum processes, graphite remains the undisputed, cost-effective champion.
Choosing the right material is about understanding the physics of your goal. For processes demanding extreme heat, unwavering purity, and absolute control, the quiet strength of graphite is the most elegant solution. Ensuring your furnace is designed by experts who master these material properties is critical to achieving your desired outcomes.
If your work demands this level of precision and control, we can help you build the ideal thermal environment. Contact Our Experts
Visual Guide
Related Products
- Vacuum Hot Press Furnace Machine Heated Vacuum Press Tube Furnace
- Vacuum Hot Press Furnace Machine Heated Vacuum Press
- 600T Vacuum Induction Hot Press Vacuum Heat Treat and Sintering Furnace
- 2200 ℃ Tungsten Vacuum Heat Treat and Sintering Furnace
- Vacuum Heat Treat Sintering Furnace with Pressure for Vacuum Sintering
Related Articles
- How Vacuum Hot Press Furnaces Transform Advanced Material Engineering
- Why Your High-Temperature Materials Fail: The Hidden War Inside Your Furnace
- The Pressure Spectrum: Why a Vacuum Furnace's Capability Isn't a Single Number
- Beyond Heat: The Art of Material Purity in Vacuum Furnaces
- Is Your Material Data Real? Why Temperature Gradients Are Sabotaging Your Cu2Se Resistivity Measurements




















