Sealing quartz ampoules at a vacuum level of 10^-5 mbar is a fundamental requirement to guarantee the complete exclusion of atmospheric air and moisture from the reaction vessel. This specific vacuum threshold is technically necessary to prevent the oxidation of raw materials and inhibit undesirable side reactions that would otherwise occur during the high-temperature growth phase of Chemical Vapor Transport (CVT).
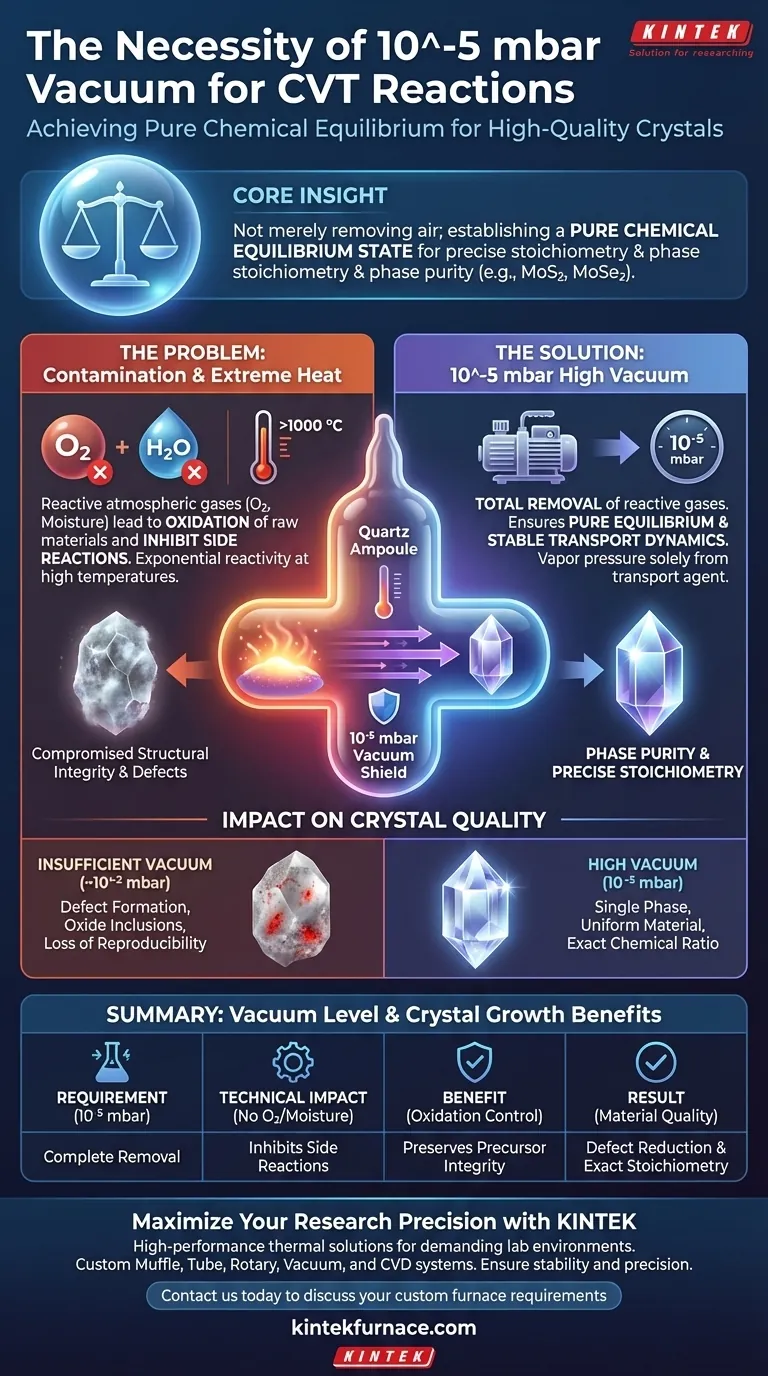
Core Insight A vacuum of 10^-5 mbar is not merely about removing air; it is about establishing a pure chemical equilibrium state. This contaminant-free environment is the primary determinant in achieving the precise stoichiometry and phase purity required for high-quality crystals like MoS2 and MoSe2.

The Role of Vacuum in Chemical Purity
Complete Elimination of Contaminants
The primary technical function of a 10^-5 mbar vacuum is the total removal of reactive atmospheric gases. Even trace amounts of oxygen or moisture trapped within the ampoule can react aggressively with the source materials.
Preventing Oxidation
At the molecular level, residual oxygen leads to the immediate oxidation of raw materials. This degrades the precursor material before the transport reaction even begins, effectively ruining the feedstock.
Inhibiting Side Reactions
Without a high-vacuum seal, moisture acts as a catalyst for undesirable side reactions. These competing chemical pathways produce impurities that incorporate into the growing crystal lattice, compromising its structural integrity.
Thermodynamics at High Temperatures
Managing Extreme Heat
CVT reactions often require temperatures exceeding 1000 °C. At these thermal extremes, the reactivity of residual gases increases exponentially, making a standard low-vacuum seal insufficient for protection.
Ensuring Chemical Equilibrium
The success of CVT relies on maintaining a pure chemical equilibrium state. The presence of foreign gas molecules disrupts the thermodynamic balance required for the transport agent to effectively move material from the source to the sink.
Stable Transport Dynamics
A 10^-5 mbar environment ensures that the vapor pressure inside the ampoule is generated solely by the transport agent and the source material. This isolation allows for predictable and controlled transport rates.
Impact on Crystal Quality
Achieving Phase Purity
High-vacuum sealing is the critical factor in achieving phase purity in the final product. By eliminating variables that cause foreign phases to nucleate, the growth process yields a single, uniform material.
Precise Stoichiometry
For complex materials like MoS2 and MoSe2, the ratio of elements (stoichiometry) must be exact. The ultra-low pressure environment prevents the loss of volatile elements to oxidation, ensuring the final crystal matches the intended chemical formula exactly.
The Risks of Insufficient Vacuum
Compromised Structural Integrity
If the vacuum level is insufficient (e.g., only 10^-2 or 10^-3 mbar), the resulting crystals often suffer from defect formation. These defects arise from oxide inclusions that disturb the atomic lattice.
Loss of Reproducibility
Failing to achieve 10^-5 mbar introduces an uncontrolled variable into the experiment. This makes it impossible to reliably reproduce high-quality growth runs, as the level of contamination will vary between ampoules.
Achieving Precision in Crystal Growth
To ensure the success of your Chemical Vapor Transport reactions, align your sealing protocols with your specific material goals:
- If your primary focus is Phase Purity: You must verify that your vacuum system reliably reaches 10^-5 mbar to completely eliminate the risk of foreign phase nucleation.
- If your primary focus is Precise Stoichiometry: You must treat the sealing process as a critical control point to prevent oxidation from altering the chemical ratio of your MoS2 or MoSe2 crystals.
Ultimately, the integrity of your vacuum seal dictates the integrity of your crystal.
Summary Table:
| Requirement | Technical Impact | Benefit to Crystal Growth |
|---|---|---|
| Vacuum Level | 10^-5 mbar threshold | Complete removal of atmospheric moisture and O2 |
| Oxidation Control | Inhibits side reactions | Preserves precursor integrity and feedstock purity |
| Thermodynamics | Pure equilibrium state | Predictable transport rates and stable dynamics |
| Material Quality | Defect reduction | Achievement of exact stoichiometry (e.g., MoS2, MoSe2) |
Maximize Your Research Precision with KINTEK
Don't let atmospheric contaminants compromise your material synthesis. KINTEK provides high-performance thermal solutions designed for the most demanding lab environments. Backed by expert R&D and manufacturing, we offer a comprehensive range of Muffle, Tube, Rotary, Vacuum, and CVD systems, all fully customizable to meet your unique high-temperature research needs.
Whether you are growing high-purity crystals or conducting complex chemical vapor transport, our equipment ensures the stability and precision your work deserves.
Contact us today to discuss your custom furnace requirements and see how KINTEK can enhance your lab's efficiency and results.
Visual Guide
References
- Bhupendra Mor, Kirti Korot. Comparative optical response and structural assessment of MoS₂ and MoSe₂ single crystals grown via iodine-assisted chemical vapor transport. DOI: 10.33545/26647575.2025.v7.i2a.168
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
People Also Ask
- Why is a stainless steel autoclave with a Teflon liner necessary for BiVO4? Ensure Purity & High Performance
- Why use a stainless steel autoclave with a PTFE liner for Ce-MOF synthesis? Ensure Safety and Purity
- Why is a PTFE-lined stainless steel autoclave used for Ni12P5 synthesis? Key Benefits for Nanomaterial Production
- Why is a copper getter chamber integrated into heating systems? Ensure Ultra-Pure Alloy Processing
- How does a Mass Flow Controller influence 2D superlattices? Precision CVD Control for Sub-10nm Patterning
