The Furnace's Dilemma
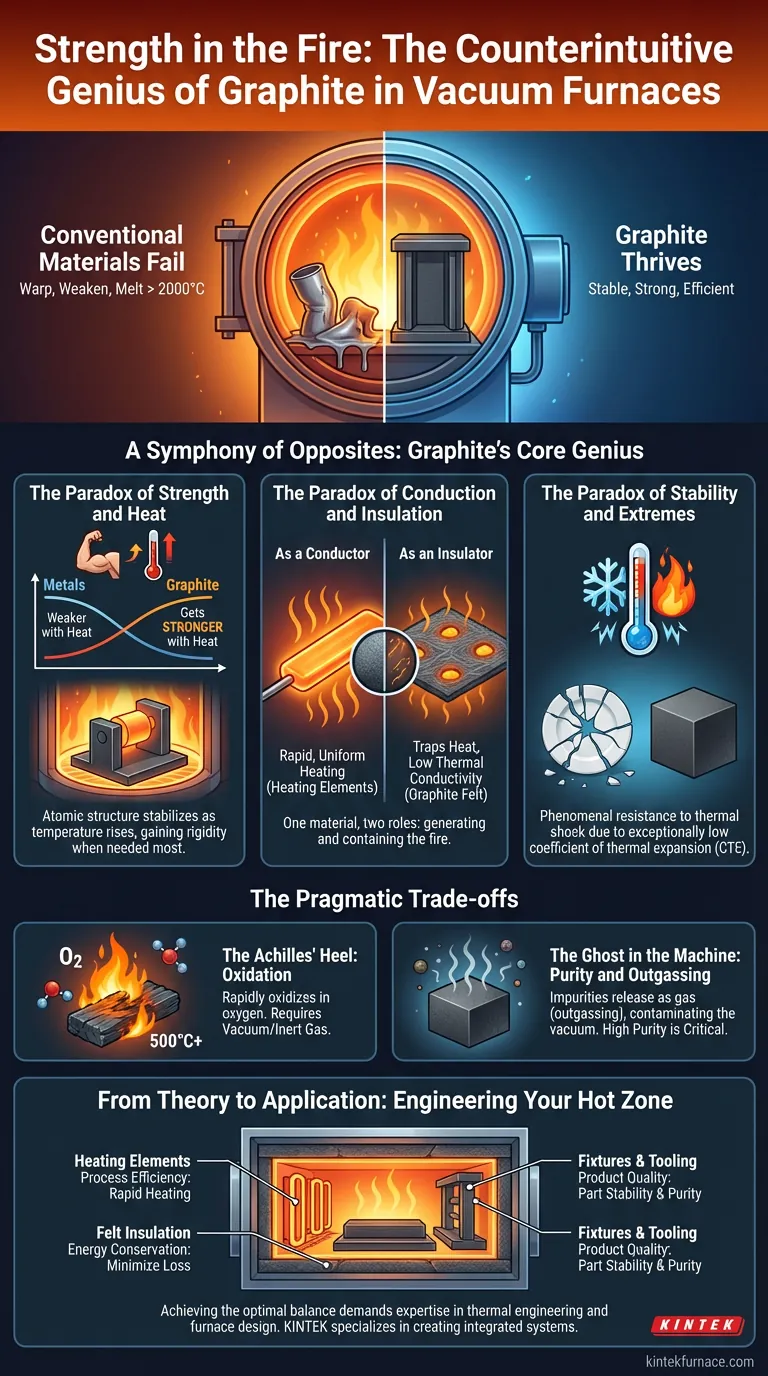
Imagine the engineering challenge: design a chamber that can contain temperatures hot enough to alter the very structure of metals, often exceeding 2000°C. Now, do it in a near-perfect vacuum, an environment where oxygen, the typical catalyst for high-temperature failure, is absent.
In this hostile world, conventional materials fail spectacularly. Metals warp, weaken, and melt. Ceramics, while heat-resistant, can crack under the immense stress of rapid temperature changes.
The problem isn't just finding a material that can survive. It's finding one that thrives—a material whose properties align perfectly with the brutal physics of the environment. The solution, elegantly simple and profoundly effective, is graphite.
A Symphony of Opposites: Graphite's Core Genius
The choice of graphite isn't based on a single virtue but on a synergy of seemingly contradictory properties. It behaves in ways that are counterintuitive, making it the ideal architect for the furnace's "hot zone."
The Paradox of Strength and Heat
Most materials we encounter follow a simple rule: as they get hotter, they get weaker. Metals lose their tensile strength and begin to creep.
Graphite defies this rule.
As its temperature rises, graphite's atomic structure becomes more stable, and it actually gets stronger. This incredible property means that fixtures, racks, and structural elements inside the furnace don't just endure the heat; they gain rigidity precisely when it's needed most, ensuring the workpieces they hold remain stable and uncontaminated.
The Paradox of Conduction and Insulation
A material is typically either a good conductor of heat or a good insulator. It's rare for one substance to excel at both.
Yet, graphite does exactly that.
- As a Conductor: In its solid, dense form, graphite has high thermal conductivity. This makes it a perfect material for heating elements, allowing for the rapid and uniform transfer of energy to heat the chamber efficiently.
- As an Insulator: When processed into a fibrous mat known as graphite felt, its structure changes. The fibers create countless tiny pockets that trap heat, making it an exceptionally effective, lightweight insulator.
This duality is a masterpiece of material science. The very same element used to generate the fire is also used to contain it.
The Paradox of Stability and Extremes
Extreme temperature swings create immense internal stress in materials. This thermal shock can cause brittle materials like ceramics to shatter.
Graphite possesses an exceptionally low coefficient of thermal expansion (CTE). It barely expands or contracts when subjected to radical temperature changes. This inherent "calmness" gives it phenomenal resistance to thermal shock, preventing the warping and cracking that would destroy lesser materials.
The Pragmatic Trade-offs
No material is perfect. Graphite's dominance in vacuum furnaces is contingent on understanding and respecting its primary limitation: its relationship with oxygen.
The Achilles' Heel: Oxidation
Graphite's strength exists in an oxygen-free world. Introduce oxygen at temperatures above ~500°C, and graphite will rapidly oxidize—essentially, it burns away.
This is precisely why it is the material of choice for vacuum and inert gas furnaces. The controlled atmosphere isn't just for protecting the workpiece; it's fundamental to protecting the furnace itself. The environment and the material exist in a symbiotic relationship.
The Ghost in the Machine: Purity and Outgassing
Low-quality graphite can contain trapped moisture and impurities. When heated in a vacuum, these contaminants are released in a process called outgassing, which can ruin the purity of the vacuum and contaminate sensitive products.
This is why the purity grade of the graphite is critical. For processes like semiconductor manufacturing or medical implant treatment, only high-purity, properly prepared graphite components will suffice.
From Theory to Application: Engineering Your Hot Zone
Designing an effective hot zone is a system-level challenge. The choice of graphite components is a strategic decision that directly impacts performance, efficiency, and final product quality.
| Strategic Goal | Key Graphite Component & Property |
|---|---|
| Process Efficiency | Heating Elements: High thermal conductivity for rapid, uniform heating rates. |
| Energy Conservation | Felt Insulation: Low thermal conductivity to minimize heat loss and reduce power consumption. |
| Product Quality | Fixtures & Tooling: High-purity, precisely machined components to ensure part stability and prevent contamination. |
Achieving the optimal balance between these elements requires more than just high-quality materials; it demands expertise in thermal engineering and furnace design. Companies like KINTEK, with deep R&D and manufacturing capabilities, specialize in creating these integrated systems. Their ability to customize everything from CVD systems to vacuum furnaces ensures the graphite architecture is perfectly tuned to the specific process requirements.
Graphite is more than just a refractory material; it is a complete, elegant solution to the hostile environment of a vacuum furnace. Understanding its paradoxical nature is the key to unlocking new levels of performance and precision in high-temperature applications.
Whether you are developing a new process or optimizing an existing one, the performance of your furnace is defined by the quality and design of its core. To engineer a system that perfectly matches your unique needs, Contact Our Experts.
Visual Guide
Related Products
- Vacuum Hot Press Furnace Machine Heated Vacuum Press Tube Furnace
- Vacuum Hot Press Furnace Machine Heated Vacuum Press
- 600T Vacuum Induction Hot Press Vacuum Heat Treat and Sintering Furnace
- 2200 ℃ Tungsten Vacuum Heat Treat and Sintering Furnace
- Vacuum Heat Treat Sintering Furnace with Pressure for Vacuum Sintering
Related Articles
- The Pressure Spectrum: Why a Vacuum Furnace's Capability Isn't a Single Number
- How Vacuum Hot Press Furnaces Transform Advanced Material Engineering
- The Unseen Enemy: Why Vacuum Furnaces Are a Bet on Perfection
- How to Select the Right Vacuum Hot Pressing Furnace Temperature for Your Materials
- The Physics of Perfection: How Vacuum Furnaces Eliminate Metallurgical Uncertainty




















