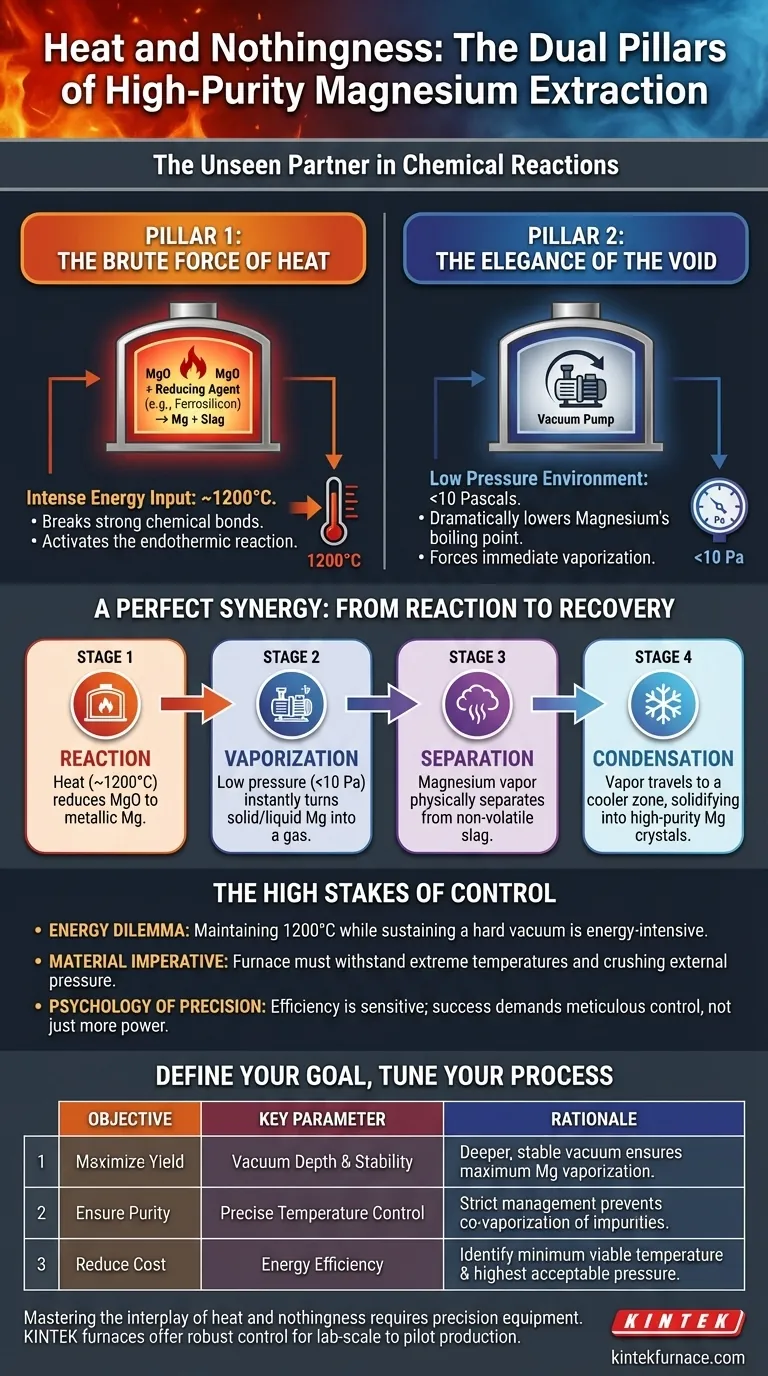
The Unseen Partner in Chemical Reactions
We have a natural bias for the visible. In high-temperature chemistry, we see the glow of the heating elements, the molten slag, the finished metal. We focus on what we add—heat, raw materials, reducing agents.
But often, the most powerful force in a system is what we take away.
Extracting metallic magnesium from ores like szaibelyite isn't just a matter of "cooking" it hot enough. That’s only half the story. The true elegance of the process lies in mastering an invisible partner: the vacuum. The furnace isn't just a heater; it's an environment manipulator that fundamentally bends the laws of physics to its will.
Pillar 1: The Brute Force of Heat
First, the obvious part. The conversion of magnesium oxide (MgO) into metallic magnesium (Mg) is an endothermic reaction. It won't happen without a massive and continuous supply of energy.
A vacuum thermal reduction furnace provides this energy, pushing the raw material and a reducing agent like ferrosilicon to temperatures around 1200°C. This intense heat is the activation energy, the brute force required to break strong chemical bonds and initiate the reaction.
This is the chemical foundation. Without it, nothing happens. But heat alone is insufficient because it creates a new problem: how do you separate the freshly made magnesium from the residual slag?
Pillar 2: The Elegance of the Void
This is where the genius of the process reveals itself. At normal atmospheric pressure, magnesium boils at over 1090°C, a temperature perilously close to the reaction temperature itself. Separating it would be messy and inefficient.
But the furnace’s vacuum pumps change the rules of the game.
By reducing the internal pressure to less than 10 Pascals—a near-perfect vacuum—the furnace dramatically lowers the boiling point of magnesium. It’s the same principle that makes water boil at a lower temperature on a mountaintop, taken to an industrial extreme.
We aren't just facilitating a chemical reaction; we are changing a fundamental physical property of the product to make it cooperate.
A Perfect Synergy: From Reaction to Recovery
These two pillars don't work in sequence; they work in perfect synergy. The moment a magnesium atom is formed by the heat-driven reaction, the vacuum environment forces it to do something extraordinary: it immediately vaporizes.
This creates a simple yet profound method of purification:
- Reaction: Intense heat (~1200°C) reduces MgO to metallic Mg.
- Vaporization: The low pressure (<10 Pa) instantly turns the solid/liquid Mg into a gas.
- Separation: This magnesium vapor physically separates from the non-volatile solid and liquid slag.
- Condensation: The vapor travels to a cooler, designated condensation zone within the furnace, where it solidifies into high-purity magnesium crystals.
This elegant dance of physics and chemistry allows for a continuous, self-purifying extraction cycle. The design of the furnace is paramount; its ability to maintain distinct thermal zones is what makes this clean separation possible.
The High Stakes of Control
This process, while elegant, is unforgiving. It operates at the extremes of temperature and pressure, creating significant engineering and operational challenges.
The Energy Dilemma
Maintaining 1200°C while simultaneously powering pumps to sustain a hard vacuum is a thermodynamic battle. It is incredibly energy-intensive and represents a primary operational cost. The system is constantly fighting to equalize, and any inefficiency costs money and affects output.
The Material Imperative
The furnace itself is a marvel of material science. It must be constructed from materials that can withstand extreme temperatures without degrading, all while resisting the crushing force of nearly 100,000 Pascals of external atmospheric pressure. The smallest leak, the slightest material failure, and the vacuum collapses, ruining the entire batch.
The Psychology of Precision
The efficiency of this process is acutely sensitive to fluctuations. There's a human tendency to believe "more is better"—a deeper vacuum or a higher temperature must lead to higher yields. But the reality is a delicate balance. Too much heat can cause impurities to vaporize along with the magnesium, compromising purity. An unstable vacuum can lead to an incomplete reaction. Success demands not just power, but meticulous control.
This is where the quality of the equipment becomes non-negotiable. A furnace isn’t just a box that gets hot; it's a precision instrument. A customizable system from a specialist like KINTEK is designed to provide that exact, stable control. Their expertise in Muffle, Tube, and Vacuum furnaces ensures the thermal uniformity and vacuum integrity required to navigate these narrow operational windows.
Define Your Goal, Tune Your Process
Optimizing the furnace's operation depends entirely on your strategic objective. There is no single "best" setting, only the best setting for your goal.
| Objective | Key Parameter to Control | The Rationale |
|---|---|---|
| Maximize Yield | Vacuum Depth & Stability | A deeper, more stable vacuum ensures the maximum amount of magnesium vaporizes for collection. |
| Ensure Purity | Precise Temperature Control | Strict temperature management prevents co-vaporization of impurities with similar vapor pressures. |
| Reduce Cost | Energy Efficiency | Identify the minimum viable temperature and highest acceptable pressure that still achieves target separation. |
Mastering this process is about understanding and controlling the interplay between heat and nothingness. The equipment you use must provide the precision to walk this fine line. With robust R&D and custom manufacturing capabilities, KINTEK's furnaces are built to meet the unique demands of your materials and production targets, whether for lab-scale research or pilot production.
To achieve the purity and yield your work demands, you need a furnace that is more than just powerful—it must be precise. Contact Our Experts
Visual Guide
Related Products
- Vacuum Hot Press Furnace Machine Heated Vacuum Press Tube Furnace
- Vacuum Hot Press Furnace Machine Heated Vacuum Press
- 600T Vacuum Induction Hot Press Vacuum Heat Treat and Sintering Furnace
- 2200 ℃ Tungsten Vacuum Heat Treat and Sintering Furnace
- Vacuum Heat Treat Sintering Furnace with Pressure for Vacuum Sintering
Related Articles
- The Pressure Spectrum: Why a Vacuum Furnace's Capability Isn't a Single Number
- How to Select the Right Vacuum Hot Pressing Furnace Temperature for Your Materials
- The Cascading Logic of Safety: Rethinking the Modern Vacuum Furnace
- Beyond the Parts List: The Real Reason Your Vacuum Hot Press Fails (And How to Fix It)
- How Vacuum Hot Press Furnaces Transform Advanced Material Engineering




















