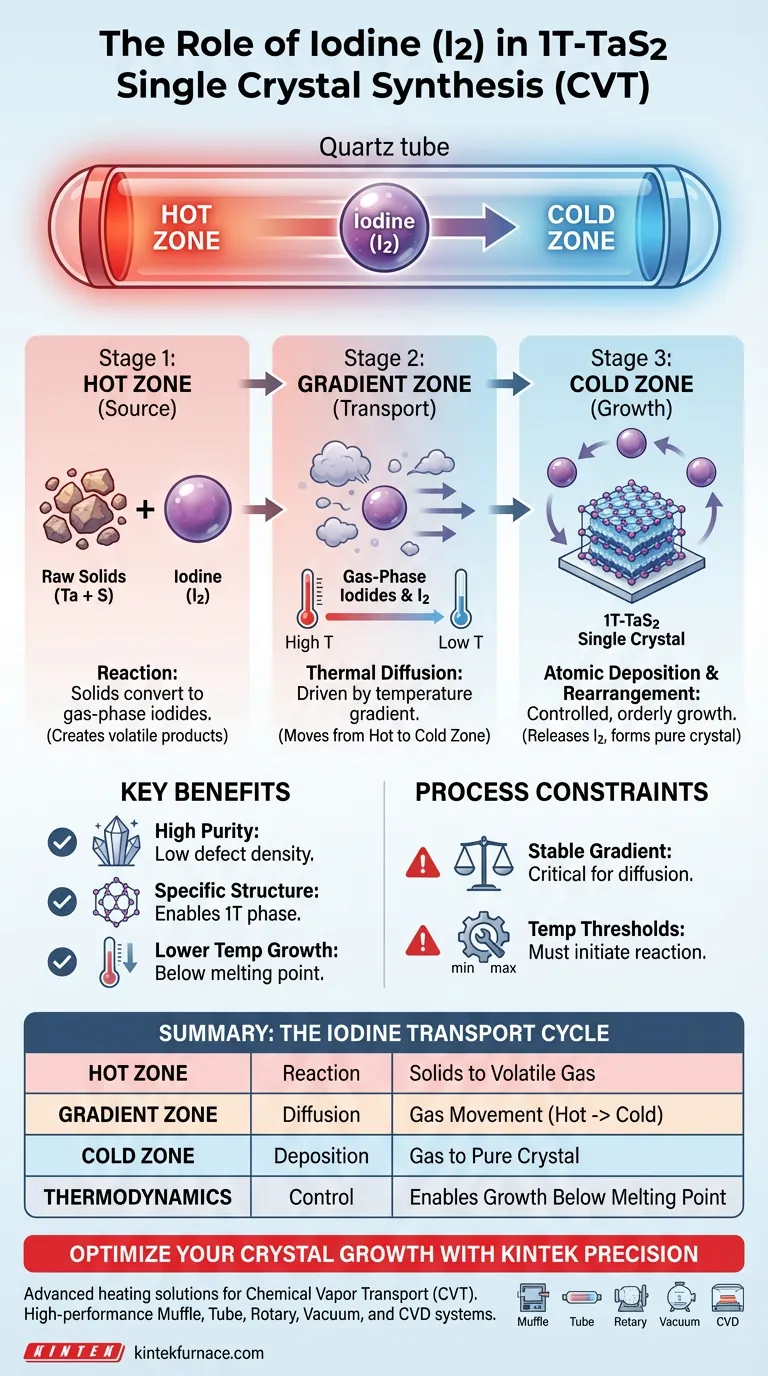
Iodine (I2) serves as the fundamental vehicle for material transfer in the synthesis of 1T-TaS2 single crystals. It functions as a chemical transport agent that reacts with solid raw materials at high temperatures to create volatile gas-phase products. These gases are then driven by a thermal gradient to deposit purified crystal layers in a cooler zone.
By converting solids into gas and back again, iodine facilitates an atomic-level rearrangement of components. This mechanism is essential for producing high-purity 1T-TaS2 crystals with low defect densities and specific crystalline structures.
The Mechanism of Chemical Transport
To understand why iodine is used, you must look beyond the simple movement of material. The value lies in the phase transition and the control it affords over crystal growth.
Reacting at the Source
In the high-temperature zone (often called the hot end), solid raw materials interact chemically with iodine.
This reaction converts the solid precursors into gas-phase iodides. Without this conversion, the raw materials would remain stationary and unable to migrate effectively within the quartz tube.
Driven by Thermodynamics
Once the materials are in the gas phase, they are subject to the thermal dynamics of the system.
A specific temperature gradient is established within the tube. This gradient creates the driving force that pushes the gas-phase components to diffuse from the hot zone toward the lower-temperature zone (the cold end).
Atomic Precision and Growth
The true benefit of this process occurs at the destination (the cold end).
As the gas reaches the cooler zone, it releases the elemental components. This allows for a controlled, atomic-level rearrangement.
This orderly deposition is what enables the growth of 1T-TaS2 single crystals that possess high integrity and specific structural phases.
Understanding the Process Constraints
While iodine transport is effective, it relies heavily on precise environmental conditions.
Dependency on Temperature Gradients
The system is entirely dependent on the stability of the temperature difference between the hot and cold zones.
If the gradient is insufficient, the gas-phase diffusion slows or halts. This prevents the material from reaching the growth zone, stopping crystal formation.
The Role of Temperature Thresholds
This method allows for crystal growth at temperatures significantly lower than the melting point of the material itself.
However, the temperature must still be high enough to initiate the initial reaction between the iodine and the raw solids. If the source temperature is too low, the gas-phase iodides will never form.
Making the Right Choice for Your Goal
The use of iodine is a strategic choice to prioritize crystal quality over simple bulk synthesis.
- If your primary focus is Purity: Ensure the temperature gradient is strictly controlled to allow for slow, deliberate atomic rearrangement, which minimizes defects.
- If your primary focus is Structure: Rely on the iodine transport mechanism to facilitate the specific atomic stacking required for the 1T phase, rather than faster, less controlled melt methods.
Iodine transforms a chaotic mixture of raw solids into a structured, high-integrity single crystal through the precision of gas-phase transport.
Summary Table:
| Stage | Action | Purpose |
|---|---|---|
| Hot Zone (Source) | Reaction with Iodine | Converts solid precursors into volatile gas-phase iodides |
| Gradient Zone | Thermal Diffusion | Drives gas-phase components from hot zone toward cold zone |
| Cold Zone (Growth) | Atomic Deposition | Facilitates atomic-level rearrangement for low-defect crystal growth |
| Thermodynamics | Temperature Control | Enables synthesis below melting points via stable thermal gradients |
Optimize Your Crystal Growth with KINTEK Precision
Successful Chemical Vapor Transport (CVT) requires absolute control over temperature gradients and environmental stability. KINTEK provides the advanced heating solutions necessary for high-integrity material synthesis. Backed by expert R&D and manufacturing, we offer high-performance Muffle, Tube, Rotary, Vacuum, and CVD systems, all of which are customizable to meet your specific research needs.
Ensure the purity and structural integrity of your 1T-TaS2 crystals with equipment designed for precision. Contact our experts today to find the perfect furnace for your lab!
Visual Guide
References
- Yihao Wang, Liang Cao. Dualistic insulator states in 1T-TaS2 crystals. DOI: 10.1038/s41467-024-47728-0
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- Custom Made Versatile CVD Tube Furnace Chemical Vapor Deposition CVD Equipment Machine
- Slide PECVD Tube Furnace with Liquid Gasifier PECVD Machine
- Multi Heating Zones CVD Tube Furnace Machine for Chemical Vapor Deposition Equipment
- Split Chamber CVD Tube Furnace with Vacuum Station CVD Machine
- Small Vacuum Heat Treat and Tungsten Wire Sintering Furnace
People Also Ask
- What temperature range do standard CVD tube furnaces operate at? Unlock Precision for Your Material Deposition
- What is a CVD tube furnace and what is its primary function? Discover Precision Thin-Film Deposition
- How might AI and machine learning enhance CVD tube furnace processes? Boost Quality, Speed, and Safety
- What are the key features of CVD tube furnaces for 2D material processing? Unlock Precision Synthesis for Superior Materials
- How does CVD tube furnace sintering enhance graphene growth? Achieve Superior Crystallinity and High Electron Mobility



















