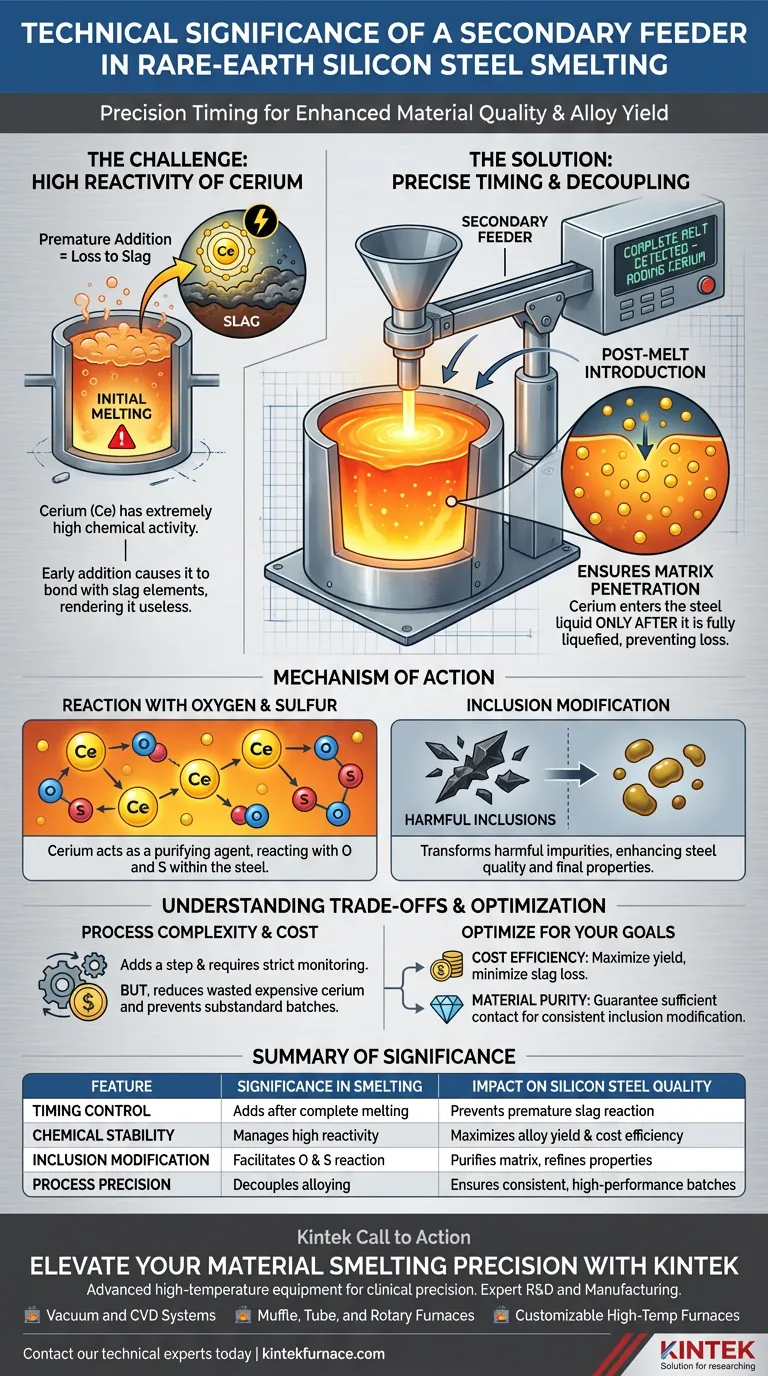
The primary technical significance of a secondary feeder is its ability to decouple the addition of volatile alloying elements from the initial melting process. In the production of rare-earth cerium modified silicon steel, this device is essential because it allows for the addition of cerium only after the base steel has completely liquefied. This precise timing mitigates the element's high chemical reactivity, preventing it from being consumed by the slag before it can effectively treat the steel.
By delaying the introduction of cerium until the melt is established, the secondary feeder ensures the element reacts with oxygen and sulfur within the steel matrix rather than being lost to waste. This is the critical step for successfully modifying non-metallic inclusions and improving the steel's final properties.

The Challenge of High Reactivity
Understanding Chemical Activity
Cerium is characterized by extremely high chemical activity. It has a strong natural tendency to bond immediately with surrounding elements.
The Risk of Premature Addition
If cerium is added at the beginning of the smelt or before the steel is fully molten, it is likely to react prematurely. This results in significant elemental loss to the slag, rendering the expensive alloy useless for its intended purpose.
The Role of Precise Timing
Post-Melt Introduction
The secondary feeder solves the reactivity problem by enabling addition at a specific, controlled moment. The reference dictates that cerium must be added strictly after the steel liquid has completely melted.
Ensuring Matrix Penetration
By adding the alloy to a fully liquid bath, the secondary feeder ensures the cerium can sufficiently enter the steel liquid. This physical integration is necessary for the chemical reactions to take place where they matter most—inside the melt, not on the surface.
Mechanism of Action
Reaction with Oxygen and Sulfur
Once successfully introduced into the molten steel, the cerium acts as a purifying agent. It targets and reacts with oxygen and sulfur present in the liquid.
Modification of Inclusions
The ultimate technical goal of this process is the effective modification of non-metallic inclusions. By controlling the reaction through the secondary feeder, the cerium transforms harmful impurities into less detrimental forms, enhancing the steel's quality.
Understanding the Trade-offs
Process Complexity vs. Material Quality
Using a secondary feeder introduces an additional step and variable to the smelting process compared to single-step batch charging. It requires strict monitoring to identify the exact moment the steel is "completely melted."
Cost Implications
However, the trade-off is heavily weighted in favor of the feeder. Without it, the "simpler" process results in unpredictable yield rates of rare-earth elements. The cost of equipment operation is offset by the reduction in wasted cerium and the prevention of substandard material batches.
Optimizing Your Smelting Process
To ensure you are getting the most out of your secondary feeder system, consider your primary production objectives:
- If your primary focus is Cost Efficiency: Utilize the secondary feeder to maximize the yield of expensive rare-earth elements, minimizing the amount lost to slag.
- If your primary focus is Material Purity: Rely on the feeder to guarantee sufficient cerium contact with oxygen and sulfur, ensuring consistent modification of non-metallic inclusions.
Precision in timing is the difference between wasted ingredients and high-performance steel.
Summary Table:
| Feature | Significance in Smelting | Impact on Silicon Steel Quality |
|---|---|---|
| Timing Control | Adds cerium only after complete melting | Prevents premature reaction with slag |
| Chemical Stability | Manages high reactivity of Cerium (Ce) | Maximizes alloy yield and cost efficiency |
| Inclusion Modification | Facilitates reaction with O and S | Purifies the steel matrix and refines properties |
| Process Precision | Decouples alloying from initial melting | Ensures consistent, high-performance material batches |
Elevate Your Material Smelting Precision with KINTEK
Don't let expensive rare-earth elements go to waste. KINTEK provides the advanced high-temperature equipment you need to achieve clinical precision in your smelting processes. Backed by expert R&D and manufacturing, we offer a comprehensive range of laboratory solutions including:
- Vacuum and CVD Systems for high-purity environments.
- Muffle, Tube, and Rotary Furnaces for uniform thermal processing.
- Customizable High-Temp Furnaces tailored to your specific silicon steel modification needs.
Ready to optimize your alloy yields and modify inclusions effectively? Contact our technical experts today to find the perfect system for your lab.
Visual Guide
References
- Lei Zhao, Xiaoyang Fu. Effect of rare-earth Ce on the texture of non-oriented silicon steels. DOI: 10.1515/htmp-2022-0321
This article is also based on technical information from Kintek Furnace Knowledge Base .
Related Products
- CF KF Flange Vacuum Electrode Feedthrough Lead Sealing Assembly for Vacuum Systems
- Multi Heating Zones CVD Tube Furnace Machine for Chemical Vapor Deposition Equipment
- High Performance Vacuum Bellows for Efficient Connection and Stable Vacuum in Systems
- Stainless Steel Quick Release Vacuum Chain Three Section Clamp
- Ultra High Vacuum CF Observation Window Flange with High Borosilicate Glass Sight Glass
People Also Ask
- What role does a Teflon-lined stainless steel autoclave play in the hydrothermal synthesis of PtLaOx@S-1 catalysts?
- Why use a stainless steel autoclave with a PTFE liner for Ce-MOF synthesis? Ensure Safety and Purity
- Why is a PTFE-lined stainless steel autoclave used for Ni12P5 synthesis? Key Benefits for Nanomaterial Production
- Why is a vacuum sealing process necessary for the synthesis of TaAs2 single crystals? Ensuring Purity in CVT Method
- How does a Mass Flow Controller influence 2D superlattices? Precision CVD Control for Sub-10nm Patterning



















