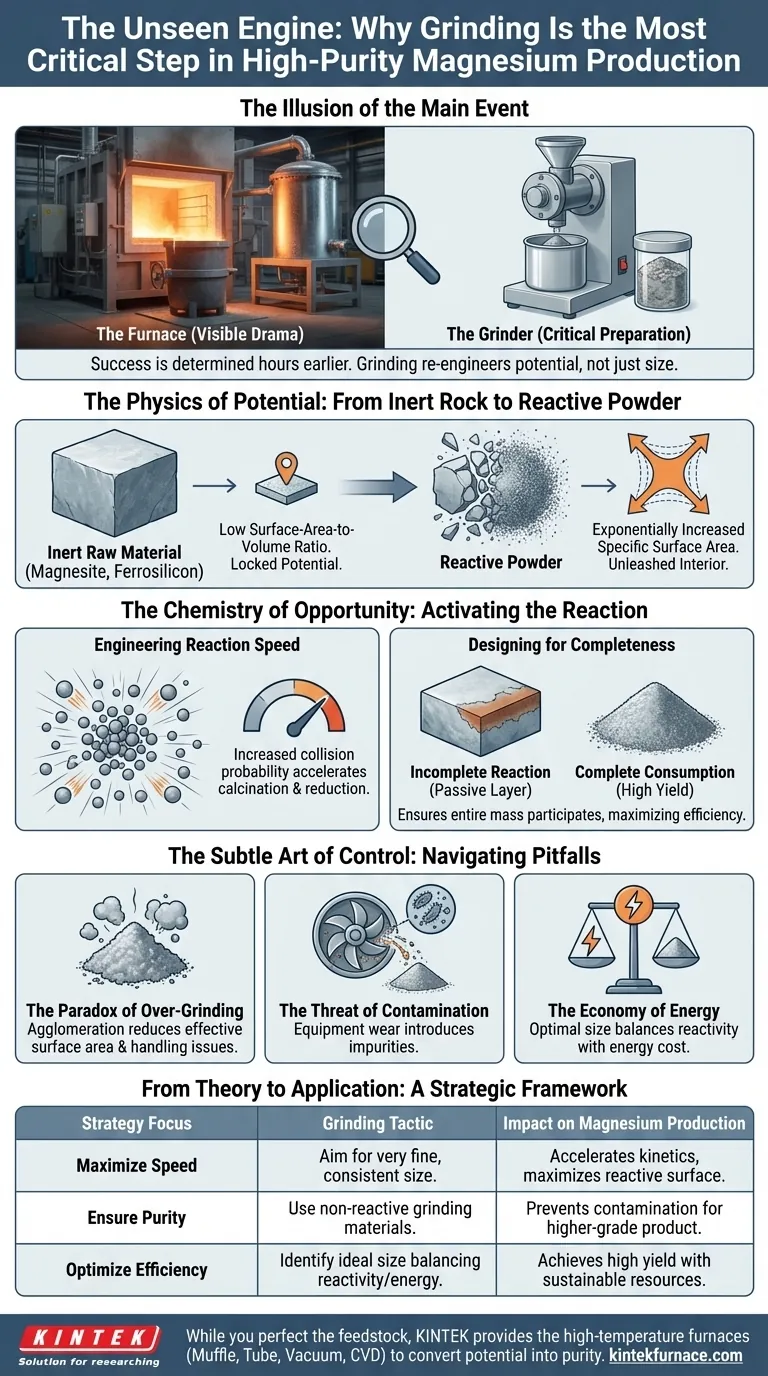
The Illusion of the Main Event
In any high-temperature experiment, our attention is instinctively drawn to the fire. We focus on the furnace, the crucible glowing under immense heat, the condensation chamber where the final product emerges. This is the visible, dramatic part of the process.
But the success or failure of that process was likely determined hours earlier, in a far less glamorous step: the simple act of grinding the raw materials.
It's a common psychological blind spot. We fixate on the climactic event and treat the preparation as a mere logistical chore. Yet, in the world of material science, preparation isn't the prelude to the experiment; it is the experiment. Grinding is not about making materials smaller; it's about fundamentally re-engineering their potential.
The Physics of Potential: From Inert Rock to Reactive Powder
The journey of magnesium production begins with materials like magnesite and ferrosilicon alloy—solid, stable, and largely unreactive in their bulk form.
The Tyranny of the Surface
An unground piece of material is like a fortress. Its potential for chemical reaction is locked away inside, while only a tiny fraction of its atoms—those on the outermost surface—are exposed. The surface-area-to-volume ratio is punishingly low. The reaction plods along, slow and inefficient, limited by this physical bottleneck.
Unleashing the Interior
Grinding shatters this fortress. It is a controlled, mechanical act of liberation. With each fracture, new surfaces are created, exponentially increasing the specific surface area—the total reactive area exposed per unit of mass.
This isn't just a physical change. It's the transformation of an inert lump into a highly reactive powder, a feedstock where nearly every particle is ready and available for the chemical transformation to come.
The Chemistry of Opportunity: Activating the Reaction
This newly created surface area directly governs the two most important factors in chemical kinetics: speed and completeness.
Engineering Reaction Speed
A chemical reaction is a game of probability. It relies on the collision of reactant molecules. By increasing the surface area, you are not just creating more contact points; you are radically increasing the probability of successful collisions at any given moment.
The result is a dramatic acceleration in the calcination and reduction reactions that produce magnesium vapor. The process becomes faster not through more heat or pressure, but through better design at the atomic level.
Designing for Completeness
A larger reactive surface also ensures the reaction goes to completion. In bulk materials, the reaction can form a passive layer on the surface, preventing the inner material from ever participating. This leaves valuable reactants unused, lowering the final yield.
A fine, uniform powder ensures that the entire mass of the feedstock is consumed, maximizing efficiency and creating the ideal precursor for a pure, consistent end product.
The Subtle Art of Control: Navigating the Pitfalls of Preparation
While essential, the grinding process is not a matter of brute force. It's a delicate balancing act, where oversimplification can introduce new, and sometimes worse, problems.
- The Paradox of Over-Grinding: Grinding too finely can be counterproductive. Ultra-fine powders can clump together (agglomerate), which ironically reduces the effective surface area you fought so hard to create. They also pose handling and safety challenges.
- The Threat of Contamination: The grinding equipment itself can be a silent saboteur. Microscopic fragments from milling surfaces can break off and mix with your powder, introducing impurities that compromise the purity of your final magnesium.
- The Economy of Energy: Grinding is an energy-intensive process. The goal is not the finest possible powder, but the optimal powder size—one that provides excellent reactivity without wasting excessive energy and time.
From Theory to Application: A Strategic Framework
The right approach to grinding depends entirely on the primary goal of your experiment. Your preparation strategy should be a conscious choice, not a default procedure.
| Strategy Focus | Grinding Tactic | Impact on Magnesium Production |
|---|---|---|
| Maximize Speed | Aim for a very fine, consistent particle size. | Accelerates reaction kinetics by maximizing reactive surface. |
| Ensure Purity | Use grinding equipment made from non-reactive materials. | Prevents contamination for a higher-grade final product. |
| Optimize Efficiency | Identify the ideal particle size that balances reactivity and energy cost. | Achieves high yield with a sustainable investment of time and resources. |
Mastering the "unseen" preparatory stage is what transforms a standard experiment into a controlled, repeatable, and highly successful process. The potential unlocked in the grinder is only realized in the furnace. The journey requires a system where every component is engineered for precision and control.
At KINTEK, we understand that great results come from a holistic process. While you perfect the feedstock, our customizable high-temperature furnaces—including Muffle, Tube, Vacuum, and CVD systems—provide the stable, controlled environment needed to convert that potential into purity. Let's build a system that honors every critical step of your research. Contact Our Experts
Visual Guide
Related Products
- High Temperature Muffle Oven Furnace for Laboratory Debinding and Pre Sintering
- 1200℃ Muffle Oven Furnace for Laboratory
- Mesh Belt Controlled Atmosphere Furnace Inert Nitrogen Atmosphere Furnace
- Dental Porcelain Zirconia Sintering Ceramic Vacuum Press Furnace
- Spark Plasma Sintering SPS Furnace
Related Articles
- Beyond the Alarm: The Hidden Safety Risks in Your High-Temperature Furnace
- Why Your High-Temperature Furnace Delivers Inconsistent Results—And How Geometry Is the Key to Fixing It
- Why Your High-Temperature Furnace Fails (Even When the Temperature Is Right)
- Why Your High-Temperature Experiments Fail—And How to Fix Them for Good
- Your Furnace Meets Spec, So Why Are Your Results Inconsistent?





