The Unseen Dialogue Between Materials
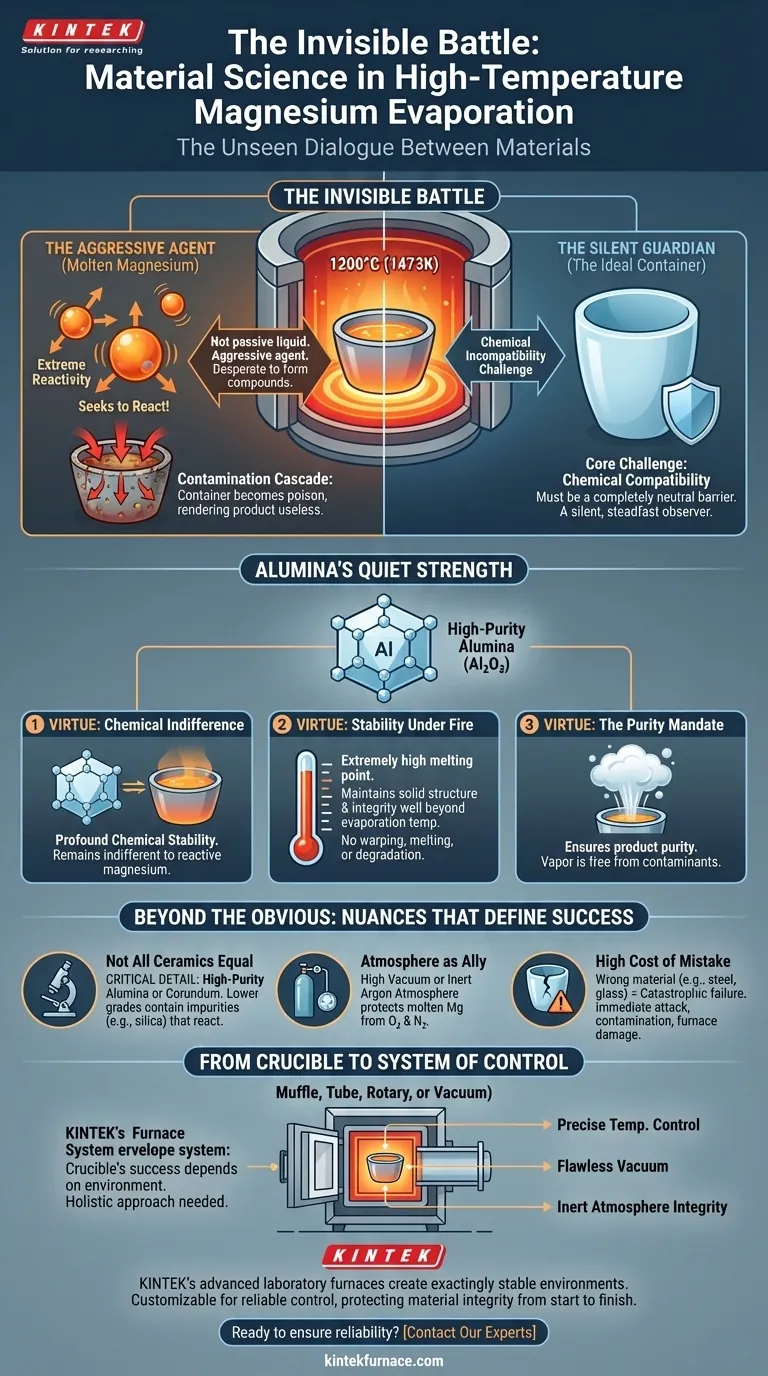
Inside a high-temperature furnace, a silent, high-stakes negotiation is taking place. At temperatures climbing towards 1200°C (1473K), a crucible holds a pool of molten magnesium. To the observer, it's a simple process of heating metal. But at the atomic level, it's a battle against chaos.
The core challenge isn't just withstanding the heat. It's a matter of chemical compatibility. Molten magnesium is not a passive liquid; it is an aggressive agent, actively seeking to react with its surroundings. The choice of its container is therefore not a trivial detail—it is the most critical decision defining the success or failure of the entire process.
The Aggressive Personality of Molten Magnesium
To engineer a solution, we must first understand the personality of the material we are trying to contain. Heat doesn't just melt magnesium; it awakens a powerful chemical reactivity.
A State of Extreme Reactivity
As magnesium transitions to its liquid state and approaches evaporation, its atoms vibrate with immense energy. This energy makes it desperate to form more stable compounds by reacting with nearly anything it touches. It will strip elements from a lesser container, dissolving the very vessel meant to hold it.
The Contamination Cascade
If the crucible reacts, it's not a minor impurity. The reaction leaches foreign elements directly into the molten magnesium. This contamination is a cascade failure, compromising the purity of the final product and rendering the entire batch useless for high-purity applications. The container becomes the poison.
The Search for an Inert Guardian
What's needed is a guardian—a material that can act as a completely neutral barrier. Its job is to hold the molten metal without participating in the chemical drama. It must be a silent, steadfast observer, ensuring that the only substance being evaporated is pure magnesium.
Alumina's Quiet Strength
This is where high-purity alumina (Al₂O₃) and similar advanced ceramics enter the scene. Their properties are a direct countermeasure to magnesium's aggressive nature.
The Virtue of Chemical Indifference
The defining characteristic of high-purity alumina is its profound chemical stability. It is exceptionally inert. Even when faced with highly reactive molten magnesium, it remains indifferent, refusing to engage in chemical reactions. This non-reactivity is its greatest strength.
Stability Under Fire
Beyond its chemical composure, alumina possesses an extremely high melting point. It maintains its solid structure and integrity well beyond the temperatures required to evaporate magnesium. It will not warp, melt, or degrade, preventing any risk of mechanical failure.
The Purity Mandate
Using an inert, stable crucible like alumina is the foundational act of ensuring product purity. When the magnesium vaporizes and is later condensed, it is free from the contaminants that a reactive container would have inevitably introduced.
Beyond the Obvious: Nuances That Define Success
Choosing "ceramic" is not enough. Success lies in understanding the subtleties of the system.
Not All Ceramics Are Created Equal
The specification is always high-purity alumina or corundum. This detail is critical. A lower-grade ceramic might contain silica or other binding agents. These impurities can themselves become reactants, defeating the entire purpose of using a ceramic crucible. Purity of the container dictates the purity of the product.
The Atmosphere as an Ally
The crucible does not work alone. High-temperature evaporation is typically conducted under a high vacuum or an inert argon atmosphere. This environment is a crucial ally, protecting the molten magnesium from reacting with oxygen or nitrogen in the air. The inert crucible and the inert atmosphere work in tandem.
The High Cost of a Simple Mistake
The consequences of choosing the wrong material are absolute. Using a steel or glass container would be catastrophic. The molten magnesium would attack it almost instantly, leading to severe contamination, a failed process, and potential damage to the furnace itself. In materials science, some choices have no margin for error.
From a Single Crucible to a System of Control
The crucible is the first line of defense, but its success depends entirely on the environment in which it operates. A high-purity alumina crucible is only as effective as the furnace that provides its operational stability.
The furnace must deliver precise temperature control, maintain a flawless vacuum, and ensure the integrity of the inert atmosphere. Any fluctuation or failure in the system undermines the perfect chemical stability of the crucible.
This is why achieving pure results requires a holistic approach. KINTEK's advanced laboratory furnaces—including Muffle, Tube, Rotary, and Vacuum systems—are engineered to create these exactingly stable environments. Customizable to your specific process, our systems provide the reliable control needed to protect material integrity from start to finish. We build the stable world your crucible needs to thrive in.
Ready to ensure the reliability of your entire thermal process? Contact Our Experts
Visual Guide
Related Products
- 1400℃ High Temperature Laboratory Tube Furnace with Alumina Tube
- 1700℃ High Temperature Laboratory Tube Furnace with Alumina Tube
Related Articles
- Beyond the Alarm: The Hidden Safety Risks in Your High-Temperature Furnace
- The Alchemy of Control: Mastering Material Properties with High-Temperature Tube Furnaces
- Why Your High-Temperature Experiments Fail: It’s Not the Heat, It’s the Atmosphere
- Why Your High-Temperature Furnace Fails: The Hidden Culprit Beyond the Cracked Tube
- Mastering the Micro-Environment: The Art and Science of the Laboratory Tube Furnace


