You’re at the final design review for a groundbreaking medical device. Years of research and millions in investment are on the line. And the integrity of the entire system—its sterility, its performance, its safety—hinges on a component that might only cost a few dollars: a PTFE seal.
You specified “medical-grade” PTFE. The datasheet looks perfect. Yet, a nagging question remains: is it truly safe? What if, under the unique stresses of your application, it leaches a microscopic contaminant? What if it deforms just enough to compromise a sterile barrier after thousands of cycles? This uncertainty is where projects stall and risks multiply.
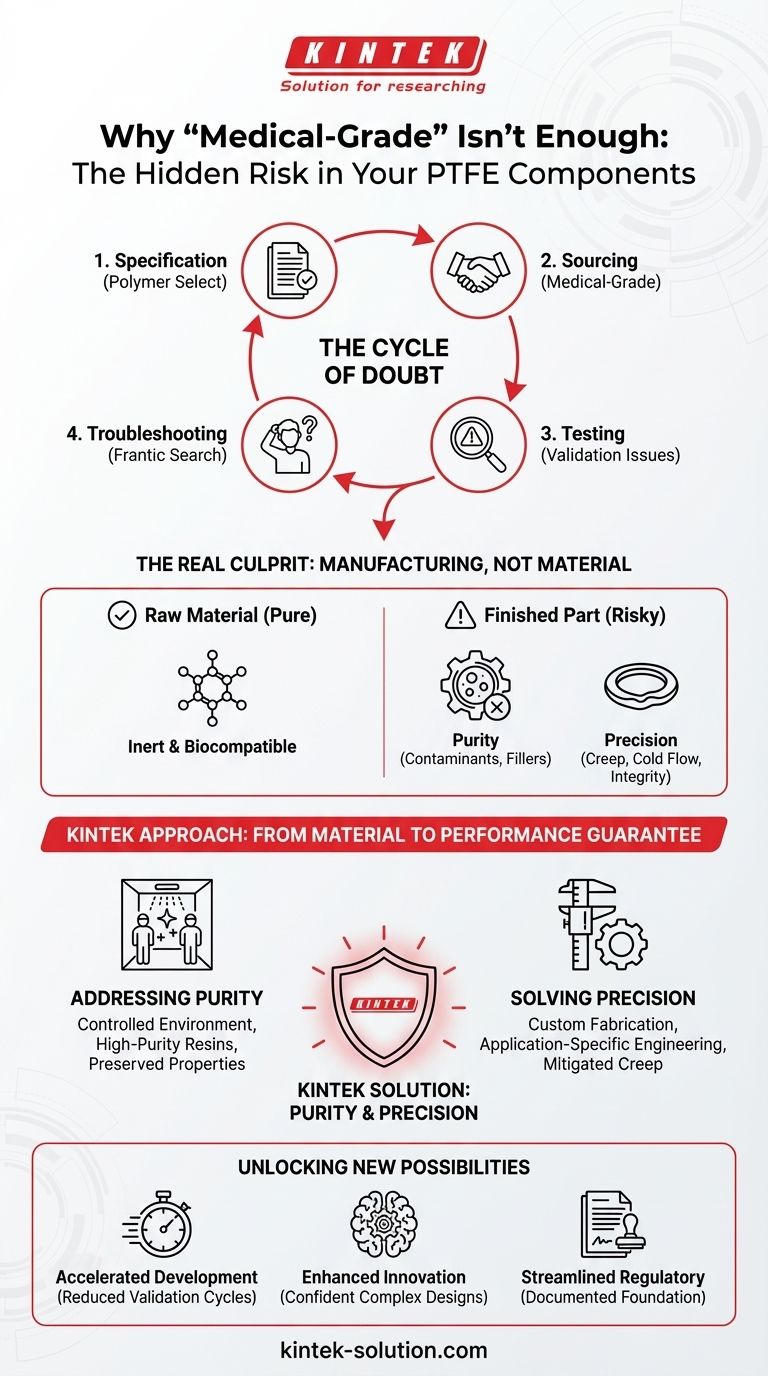
The Cycle of Doubt: Why Standard Datasheets Fall Short
This scenario is familiar to countless engineers and product managers. The challenge of material selection for a critical medical application often becomes a frustrating cycle:
- Specification: You select a polymer like PTFE for its well-known chemical inertness and biocompatibility.
- Sourcing: You procure a component that is certified as "medical-grade."
- Testing: During validation, you encounter an issue. Perhaps it’s a slight discoloration after steam autoclaving, inconsistent sealing pressure in a prototype, or an unexpected finding in an extractables and leachables (E&L) study.
- Troubleshooting: You question everything. Was it the sterilization process? A design flaw in the housing? Or was it the material itself?
The frantic search for answers begins. You scrutinize datasheets, but they only provide generic physical properties. They don't tell you about the specific batch purity, the potential for microscopic contaminants from the manufacturing line, or how the material will behave under the sustained mechanical load of your specific design.
This cycle of doubt has significant business consequences. It leads to costly project delays, budget overruns for repeated testing, and a lingering lack of confidence in the final product's long-term reliability. The cost of failure is simply too high to leave to chance.
The Real Culprit: It’s Not the Material, It's the Manufacturing
Here is the crucial turning point: The anxiety surrounding PTFE safety doesn't stem from the Polytetrafluoroethylene polymer itself. The raw material is, as science has proven, exceptionally inert and biocompatible.
The real risk lies in treating a critical component as a commodity.
The term "medical-grade" is a starting point, not a guarantee of performance. The safety and reliability of your PTFE component are determined by two factors that a standard datasheet will never reveal:
-
Purity of the Final Part: Was the component manufactured in a controlled environment? Could trace contaminants, processing aids, or fillers have been introduced during production? Pure PTFE is safe; contaminated PTFE is a liability. A certificate for the raw resin doesn't guarantee the purity of the finished, machined part.
-
Precision and Mechanical Integrity: PTFE is a relatively soft material susceptible to "creep" or "cold flow" under pressure. An off-the-shelf O-ring might meet dimensional specs, but will it maintain a reliable seal over months or years in your custom-designed housing? Failure here isn't a chemical issue; it's a mechanical one born from a lack of application-specific engineering.
This is why the common "solutions"—switching to another grade, slightly altering the design—often fail. They are attempts to fix a symptom without addressing the root cause: a disconnect between the raw material's potential and the precision of its final, manufactured form.
From Material Spec to Performance Guarantee: The KINTEK Approach
To truly eliminate this risk, you don't just need a material supplier; you need a manufacturing partner who treats every component as a piece of precision engineering. You need a process designed to bridge the gap between a raw polymer and a component you can trust with patient safety.
This is precisely why KINTEK’s approach is different. We recognize that for industries like medical and semiconductor, the value is not in the plastic, but in the purity and precision of the final part.
-
Addressing the Purity Problem: We specialize in manufacturing with high-purity PTFE resins in a controlled environment. Our process is designed to ensure that the inert properties of the raw material are preserved in the finished component you receive. We provide components, not just parts—each one backed by a deep understanding of what medical applications demand.
-
Solving the Precision Problem: A generic seal is a gamble. We provide custom fabrication, from prototype to high-volume production. This allows us to work with you to ensure the component is not only dimensionally accurate but also mechanically suited to your specific application, mitigating risks like creep and ensuring long-term sealing integrity. Our expertise isn’t just in machining PTFE; it’s in engineering PTFE solutions.
Beyond Reliability: Unlocking New Possibilities in Medical Device Design
When you eliminate the uncertainty around component purity and performance, something powerful happens. Your team is liberated from the cycle of doubt and endless troubleshooting. You can move faster, innovate with greater confidence, and focus on what you do best.
Imagine what becomes possible:
- Accelerated Development: Reduce time-consuming validation cycles by starting with components you know are pure and precisely matched to your design.
- Enhanced Innovation: Confidently design more complex, ambitious devices—from long-term implants to sophisticated diagnostic equipment—knowing your material foundation is solid.
- Streamlined Regulatory Pathways: Build your submission on a foundation of documented material purity and manufacturing precision, simplifying a critical part of the approval process.
Ultimately, partnering with a specialist like KINTEK isn't just about sourcing a better seal or liner. It's about de-risking your entire product development process and ensuring that your final device delivers on its promise of safety and efficacy.
Your project deserves more than an off-the-shelf part; it requires a tailored solution built on a foundation of expertise. Let's discuss the specific challenges of your application and how precision-fabricated PTFE components can provide the certainty you need. Contact Our Experts.
Visual Guide
Related Products
- Custom PTFE Parts Manufacturer for Teflon Parts and PTFE Tweezers
- Custom PTFE Parts Manufacturer for Teflon Containers and Components
- Custom PTFE Sleeves and Hollow Rods for Advanced Applications
- Custom PTFE Square Trays for Industrial and Laboratory Use
- Custom PTFE Teflon Balls for Advanced Industrial Applications
Related Articles
- Your "Inert" PTFE Component Might Be the Real Source of System Failure
- The Unseen Workhorse: Why PTFE Is the Default Choice for Impossible Problems
- Why Your High-Performance PTFE Parts Fail—And Why It's Not the Material's Fault
- How PTFE Solves Critical Industrial Challenges Through Material Superiority
- Beyond "Non-Stick": Why Your PTFE Components Fail and How to Fix It for Good